Dentin hypersensitivity: a state-of-the-art and novel approach with ozone therapy
June 14, 2017 / Categories: Digital Dentistry, Implant Dentistry
Zoi, Valentina

Gobbo, Margherita

Ottaviani, Giulia

Rupel, Katia

Poropat, Augusto

Di Lenarda, Roberto

Biasotto, Matteo
Abstract
Objective
The study evaluated the possibility of extending the use of ozone therapy in dentistry to the treatment of dentinal hypersensitivity.
Methods
The prospective study included 40 patients. All of the enrolled patients were randomized into two groups, allocating one patient to the ozone group and the subsequent one to the paint group. The patients’ responses to air stimuli were recorded using a numeric rating scale (NRS), asking each patient to express the degree of pain experienced on a scale of 0 to 10 at baseline, after 14 days and monthly for six months, after the last ozone session. The evaluation was performed by recording the post-treatment hypersensitivity according to the NRS and by completion of a questionnaire.
Results
Evaluating the trend of NRS values at the various time points within the two individual groups using the Friedman test, a gradual improvement of the symptoms was registered in the paint group (p < 0.024) and in the ozone group (p < 0.000).
Conclusion
Both therapies proved to be effective. The paint demonstrated an immediate desensitizing action, which was not observed in the treatment with ozone. However, in the long term, the paint did not ensure a significant reduction in NRS values. Treatment with ozone proved to be more effective in the maintenance of long-term results.
Keywords
Ozone, dentinal hypersensitivity, desensitization.
Introduction
Dentinal hypersensitivity is defined as a “sensation of pain that appears in response to chemical, thermal, tactile or osmotic stimuli in contact with exposed dentin, which cannot be related with other alterations or dental pathologies.”1Canadian Advisory Board on Dentin Hypersensitivity. Consensus-based recommendations for the diagnosis and management of dentin hypersensitivity.
→ J Can Dent Assoc. 2003 Apr;69(4):221–6.
Although dentinal hypersensitivity is a widespread condition, therapeutic strategies available today are not always effective or satisfactory.2Aranha AC, Pimenta LA, Marchi GM. Clinical evaluation of desensitizing treatments for cervical dentin hypersensitivity.
→ Braz Oral Res. 2009 Jul-Sep;23(3):333–9.
Today, there are three prevailing theories regarding the etiopathogenesis of dentinal hypersensitivity:
- The direct innervation theory assumes that a direct stimulation of the nerve endings occurs in response to external events that excite the processes of the odontoblastic body.3Abbinate A, Ardizzone VC. Cap 23 Trattamento della sensibilità dentinale.
→ In: Abbinate A, Ardizzone VC. Igienista orale. Teoria e pratica professionale. Milano: Edra editor. 2013 p. 437–8. This theory is not sufficiently supported: Few tests support the existence of nerve endings in the dentin surface, precisely where the dentin is more sensitive; moreover, the plexus of Raschkow does not become mature until complete eruption of the tooth. - The odontoblast receptor theory supposes, consequent to painful stresses, the release of neurotransmitter substances by odontoblasts is responsible for a reaction of the nerve endings present in the dentin that stimulates the pulp response. This theory assumes the ability of the odontoblasts to behave as nerve receptors and transmit signals to the nerves of the pulp. This theory has been discredited because the cellular matrix of the odontoblasts cannot be stimulated and produce nerve impulses; moreover, there are no synapses between the odontoblasts and nerves of the pulp.4Miglani S, Aggarwal V, Ahuja B. Dentin hypersensitivity: recent trends in management.
→ J Conserv Dent. 2010 Oct;13(4);218–24. - The hydrodynamic theory, proposed by Martin Brännström, is the most widely recognized and accredited for the pathophysiology of dentinal hypersensitivity.5Brännström M, Astroem A. A study on the mechanism of pain elicited from the dentin.
→ J Dent Res. 1964 Jul-Aug;43:619–25. This theory claims that dentinal hypersensitivity is the result of fluid movement within the tubules caused by thermal and physical changes or the presence of osmotic stimuli at the level of the exposed dentin. The fluid movement stimulates the baroreceptors and causes the nervous discharge.
From a clinical point of view, dentinal hypersensitivity is caused by:
- gingival recession;
- abrasion;
- erosion;
- abfraction;
- hypoplasia or abnormalities of enamel development.
Moreover, dentinal hypersensitivity may also have an iatrogenic origin: in conservative treatments, when dentin is exposed to an etching acid for too long or as a result of periodontal surgery (mucogingival, resective or regenerative).6Canadian Advisory Board on Dentin Hypersensitivity. Consensus-based recommendations for the diagnosis and management of dentin hypersensitivity.
→ J Can Dent Assoc. 2003 Apr;69(4):221–6.
Therapy and classificatio of desensitizing agents
The desensitizing agents currently on the market essentially have two modes of action:
- occlusion of the dentinal tubules, with consequent permeability reduction;
- reduction of the dentinal nerve fiber activity and therefore of the transmission of the pain stimulus to nerve centers.
Professional treatments include application of paints, varnishes, resins and bonding agents to occlude the dentinal tubules. The materials most frequently used are fluoride, potassium nitrate, potassium oxalate and calcium phosphate, and these generate endodontic crystallizations with consequent reduction of the movement of fluid in the tubules.7Gaffar A. Treating hypersensitivity with fluoride varnish.
→ Compend Contin Educ Dent. 1999;20(1 Suppl):27–33; quiz 35.
We report a complete list of all compounds, which could be used for desensitization:
Composite resins, glass ionomer cements, glass ionomer modified-resins and compomers: applied on the dentin-exposed surface, these ensure the occlusion of the tubules. 8Abbinate A, Ardizzone VC. Cap 23 Trattamento della sensibilità dentinale.
→ In: Abbinate A, Ardizzone VC. Igienista orale. Teoria e pratica professionale. Milano: Edra editor. 2013 p. 437–8.
Resin-based products with hydrophilic characteristics that contain hydroxyethyl methacrylate (HEMA), ammonium chloride, fluoride and glutaraldehyde: the last, used in different desensitizers, causes coagulation of the plasma and closing proteins in tubules, thus inhibiting the movement of dentinal fluid outward. The HEMA penetrates into the tubules and, after polymerization, occludes them, reducing permeability.
Portland cements based on silicate calcium, tricalcium silicate, tricalcium aluminate and tetra-calcium aluminoferrite: these have shown efficacy in reducing dentin permeability owing to the ability to produce hydroxyapatite in contact with body fluids containing phosphate.
Sealants: They contain microfillers, which occlude the tubules and create a barrier effect in the tooth.
Paints: These are generally based on synthetic resins, contain calcium fluoride and sodium fluoride, and exert a remineralizing, protective and insulating effect on thermal and chemical stimuli.9Gaffar A. Treating hypersensitivity with fluoride varnish.
→ Compend Contin Educ Dent. 1999;20(1 Suppl):27–33; quiz 35.
Varnishes: These have an obstructive and antibacterial action, contain fluorides and often chlorhexidine. They adhere to the dentin and gradually release the two active substances.
Fluorides: Fluoride acts on the hydroxyapatite crystals of enamel, transforming them into fluorapatite, with binding characteristics that are more stable and resistant to the action of an acidic pH and determine the precipitation of calcium phosphate into the tubules.
Pastes containing casein phosphopeptide and amorphous calcium phosphate: These substances favor the remineralization processes of the dental surfaces. The casein phosphopeptide maintains the calcium and phosphate in a noncrystalline amorphous state and acts as an adhesive, adhering to the hard and soft tissue of the oral cavity by slowly releasing calcium and phosphate ions, which penetrate into the enamel, forming apatite crystals.10Abbinate A, Ardizzone VC. Cap 23 Trattamento della sensibilità dentinale.
→ In: Abbinate A, Ardizzone VC. Igienista orale. Teoria e pratica professionale. Milano: Edra editor. 2013 p. 437–8.
Ozone
Ozone (O3) is a natural gaseous molecule defined as an allotropic form of oxygen. The idea of using ozone in medicine has developed slowly during the last century and was stimulated by the lack of antibiotics and the antiseptic properties of ozone. Its liquid form is able to penetrate into the tissue and mucous membranes. The effects of ozone in the organism depend on its chemico- physical characteristics, high reactivity and oxidative potential: antimicrobial, anti- inflammatory, analgesic, immunostimulant, anti-hypoxic and biosynthetic.11Sunnen GV. Ozone in medicine: overview and future directions.
→ J Adv Med. 1988 Fall;1(3):159–74. Owing to its high oxidative potential, ozone oxidizes the cellular components of bacterial cells, thereby modifying the intracellular components with consequent loss of function of organelles. When the cell membrane is damaged during this process, the cell is destroyed. In conclusion, ozone causes the lysis of the cell.
Ozone activities:
1. Antimicrobial:
- Damage to the cytoplasmic membrane
- Oxidation of intracellular components
2. Analgesic
3. Immunostimulant:
- Activation of the humoral immune system
- Immunoglobulin synthesis
4. Anti-hypoxic
5. Increased phagocyte activity
6. Activation of biological antioxidants:
- Activation of the aerobic processes (Krebs cycle, glycolysis, beta-oxidation of fatty acids)
- Increase of cellular metabolism (ribosomes and mitochondria)
7. Biostimulating:
- Synthesis of interleukins, leukotrienes and prostaglandins
- Immunoglobulin synthesis.
The main fields of application of ozone therapy in dentistry include:
- conservative treatment of carious lesions;
- nonsurgical periodontal therapy;
- oral pathology (cheilitis, ulcers, osteonecrosis);
- oral surgery;
Treatment of dentinal hyper – sensitivity with ozone therapy
Dentin consists of 76% minerals, 20% organic matrix and 10% water. The main component of the organic matrix is collagen.12Miglani S, Aggarwal V, Ahuja B. Dentin hypersensitivity: recent trends in management.
→ J Conserv Dent. 2010 Oct;13(4);218–24Since ozone reacts quickly to compounds containing double bonds (such as C = C, C = N and N = N, bonds and organic amine bonds contained in the collagen), 13Sunnen GV. Ozone in medicine: overview and future directions.
→ J Adv Med. 1988 Fall;1(3):159–74. dentin represents an ideal substrate for its action. The mechanism of oxidation of ozone is based on the direct and indirect effects of the reaction. Direct oxidation is a selective reaction during which ozone reacts rapidly with organic material containing double bonds, organic groups or amines. The indirect reaction leads instead to the production of hydroxyl radical groups, extremely unstable compounds that have an oxidative power even stronger than that of ozone. Hydroxyl radicals do not affect the inorganic tissue, but attack the organic components of the dentin.14Summerfelt ST, Hochheimer JN. Review of ozone processes and applications as an oxidizing agent in aquaculture.
→ Prog Fish Cult. 1997;59(2):94–105. These reactions, both direct and indirect, are responsible for the preferential attack of ozone on peritubular demineralized dentin, leading to an increase in the diameter of the dentinal tubules. Therefore, the use of ozone, not allowing the direct obstruction of the tubules, does not reduce the feeling of hypersensitivity; consequently, it is not expected that the use of ozone will reduce the pain sensation of dentinal hypersensitivity, at least not by means of the occlusion of the dentinal tubules. However, other studies have suggested that ozone increases the patency of the dentinal tubules, thus facilitating the entry of minerals from the saliva and desensitizing agents containing fluoride. 15Lynch E. Ozone: the revolution in dentistry.
→ Copenhagen: Quintessence. 2004. 300 p.16Bocci V, Bianchi L, Larini A. The ozone enigma in medicine. The biochemical relationship between ozone and body fluids may account for its biological, therapeutic and toxic effects.
→ Riv Ital Ossigeno-Ozonoterapia. 2003 Oct;2(2):113–20. Italian.
Study aim
The aim of the present study was to assess the effectiveness of ozone in reducing dentinal hyper sensitivity. The results obtained with ozone therapy were compared with those of an active control group, treated with a paint-on desensitizing agent.
Materials and methods
The prospective study was conducted at the Division of Oral Medicine and Pathology of the dental clinic of the Ospedale Maggiore (Trieste, Italy). All of the procedures performed were in accordance with the ethical standards of the institutional and national research committee and with the principles embodied in the Helsinki Declaration of 1975, as revised in 2013. The study included 40 patients between the ages of 21 and 85, selected according to the following criteria:
1. Inclusion criteria:
- presence of dentinal hypersensitivity;
- single or multiple dental elements;
- dental elements affected by noncarious lesions of the enamel (abrasion, erosion, demineralization, white spot);
- exposed root of a nonimplant prosthesis;
- cooperative and able to define the pain experienced according to a numeric rating scale (NRS).
2. Exclusion criteria:
- radiotherapy in the head neck region;
- xerostomia;
- carious lesions;
- Class V fillings;
All of the enrolled patients were randomized into two groups, allocating one patient to the ozone group and the subsequent one to the paint group, until the predetermined number was reached.
Study design
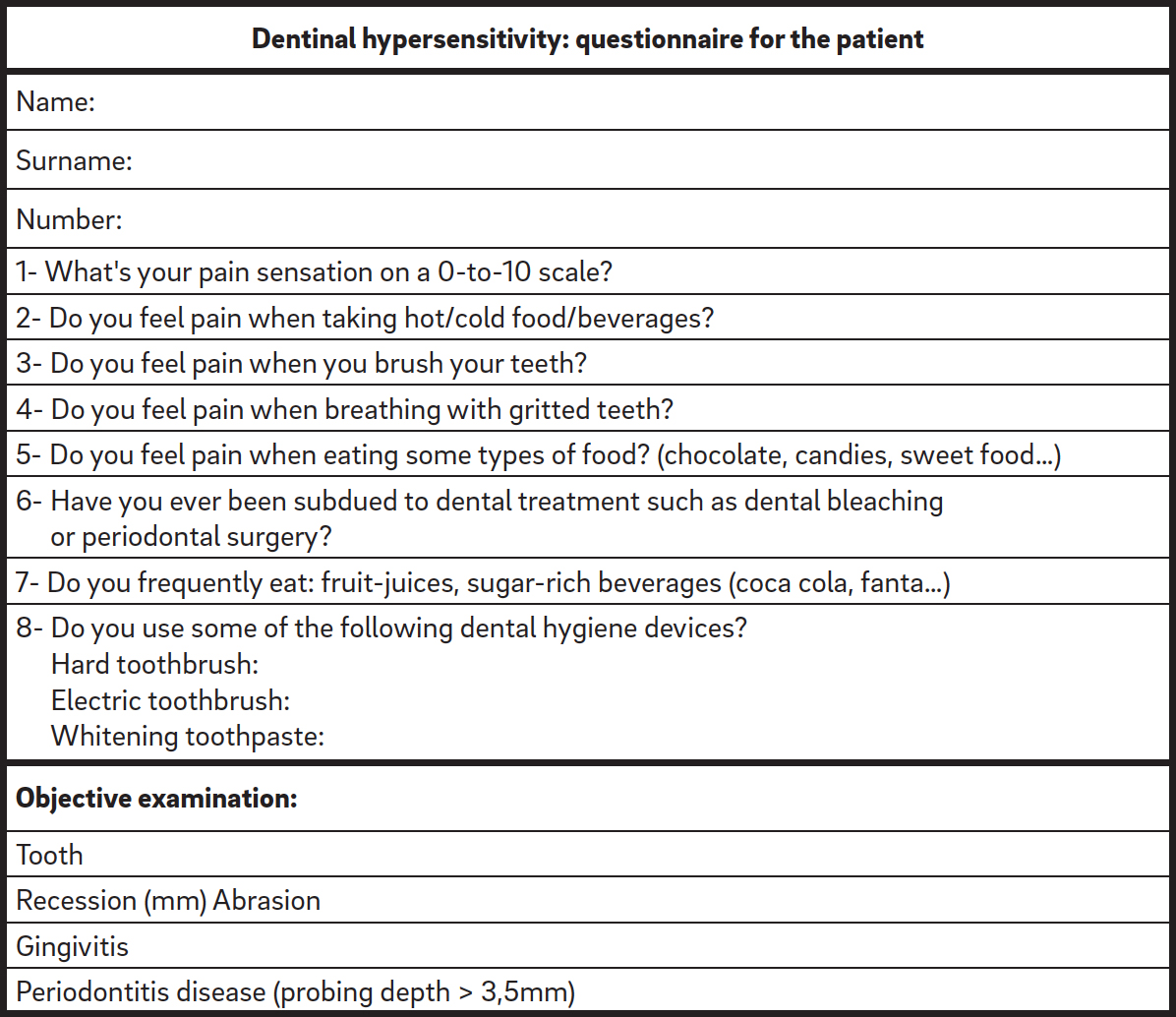
First, for all of the patients, a thorough clinical oral examination was performed in order to identify the elements affected by dentinal hypersensitivity. Then, through an objective examination, a dental hygienist recorded the presence of gingival recession and dental abrasion, performed periodontal screening and recording, and scored the patient according to the plaque index. All of the patients were asked to complete a questionnaire regarding their food and oral hygiene habits and the severity of their dentinal hypersensitivity symptoms (Appendix A).
Appendix A
Questionnaire at first visit
The pain symptomatology was quantified, evoking the stimulus through the application of a jet of compressed air at the level of the affected element. A numeric rating scale (NRS) was used, asking each patient to express the degree of pain on a scale of 0 to 10, in which 0 represented the absence of pain and 10 the worst pain imaginable.
Treatment with ozone
The ozone group was treated with the medical ozone generator OZONE DTA (Sweden & Martina, Due Carrare, Italy), which produces ozone through the formation of an electromagnetic field. Treatment with ozone was planned for four sessions one week apart each (T1, T2, T3, T4), according to the following protocol:
- evaluation and registration of the NRS (0–10) with a jet of compressed air;
- cleansing of the affected element using a dental water brush mounted on a micromotor, without using an abrasive paste;
- drying of the element with compressed air;
- ignition and activation of the machinery, set to program No. 6;
- application of the tip of the probe without contact with the dental element, but in a perpendicular position with respect to the area of the tooth concerned, with continuous movement on the whole area for a total duration of 1 min;
- evaluation and registration of the NRS (0–10) with a jet of compressed air.
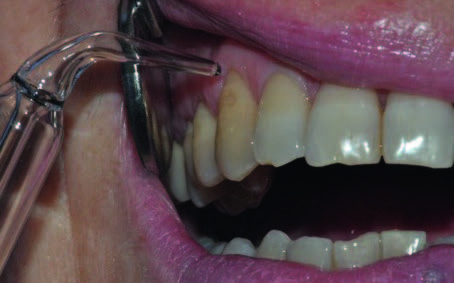
The patients were reassessed after 14 days (FU1) and monthly for six months (FU2, FU3, FU4, FU5, FU6, FU7) after the last ozone session. The evaluation was performed by recording the post-treatment hypersensitivity according to the NRS and by completion of the questionnaire. An example of a clinical case is provided in Figure 1.
- Fig. 1
Fig. 1
Ozone application at tooth #13 (affected by a vestibular recession of 3 mm).
Treatment with paint
For the paint group, the VivaSens desensitizing varnish (IvoclarVivadent, Schaan, Liechtenstein) was used. It contains ethanol, water, hydroxypropyl cellulose, potassium fluoride, polyethylene glycol dimethacrylate and other methacrylates. Treatment with VivaSens was performed in a single session, according to the following protocol:
- evaluation and registration of the NRS (0–10) with a jet of compressed air;
- cleansing of the affected element using a dental water brush mounted on a micromotor, without using an abrasive paste;
- drying of the element with compressed air;
- application of three drops of VivaSens using a microbrush over the entire surface of the element for 1 min;
- drying of the element with compressed air for 30 s;
- evaluation and registration of the NRS (0–10) with a jet of compressed air.
Once the desensitizing agent had been applied, each patient was recommended not to rinse, drink or eat for the following 30 min, as indicated by the manufacturer. The patients were reassessed, recording NRS score, after 14 days (FU1) and monthly for six months (FU2, FU3, FU4, FU5, FU6, FU7) after the last paint application.
Statistical analysis
Statistical analysis was performed using SPSS for Windows (Version 16.0; SPSS, Chicago, Ill., U.S.). A p-value of less than 0.05 was considered the cut-off of significance in the contrast of the null hypothesis. The t-test for equality of the median was used to assess the equality of the age distribution and the chi-squared test to evaluate the homogeneity of sexes in both groups. The Mann–Whitney test was used to evaluate the significance of differences between groups in NRS values at baseline (T0) and in the course of the follow-up. The Fisher two-tailed exact test was used to evaluate the homogeneity of distribution between groups regarding the following parameters: presence of recession; presence of plaque; presence of abrasion; presence of periodontal pocket depths of > 3.5 mm; pain in response to cold, hot, mechanical or osmotic stimuli; pain evoked by forced inhalation through gritted teeth; using an electric toothbrush; previous dental whitening; previous periodontal surgery; and frequent intake of acidic foods or beverages. The Friedman test was used to evaluate the variation of VAS (Visual Analog Scale) within the paint group and ozone group, respectively. The Wilcoxon test was used to assess the differences in VAS values between times within the two groups and between T0 and T1, between T0 and FU6, and between T0 and FU7 in the paint group and between T0 and T4, between T0 and FU6, and between T0 and FU7 in the ozone group, respectively. Bonferroni’s corrections were applied, where necessary, in multiple comparisons.
Results
The 40 patients, 34 females and 6 males, respectively, were distributed in two groups with an average age of 49.0 ± 17.5 (range: 22.0–85.0):
- Varnish group: 20 patients, with an average age of 50.7 ± 15.2 (range 22.0-73.0);
- Ozone group: 20 patients, with an average age of 47.3 ± 19.8 (range 22.0-85.0).
The t-test for equality of the median demonstrated that age was uniformly distributed in both groups (p < 0.547). We report the descriptive analysis with respect to the clinical examination and the questionnaire answers: – Twenty-three patients (57.5%) had at least one gingival recession of > 3 mm in the area of reported hypersensitivity.
- Twelve patients (30%) had bacterial plaque in the area of reported hypersensitivity.
- Twelve patients (30%) had abrasion in the area of reported hypersensitivity.
- Ten patients (25%) had periodontal pocket depths of > 3.5 mm at the level of the hypersensitive tooth.
- Thirty-eight patients (95%) reported pain due to cold stimuli.
- Five patients (12.5%) reported pain due to hot stimuli.
- Twenty-one patients (52.5%) reported pain due to mechanical stimuli.
- Ten patients (25%) reported pain as a result of forced inhalation through gritted teeth.
- Eight patients (20%) reported pain due to osmotic stimuli.
- Eleven patients (27.5%) reported using an electric toothbrush.
- Two patients (5%) reported having undergone a dental whitening treatment.
- Three patients (7.5%) reported having undergone periodontal surgery.
- Nine patients (22.5%) reported consuming acidic foods or beverages frequently.
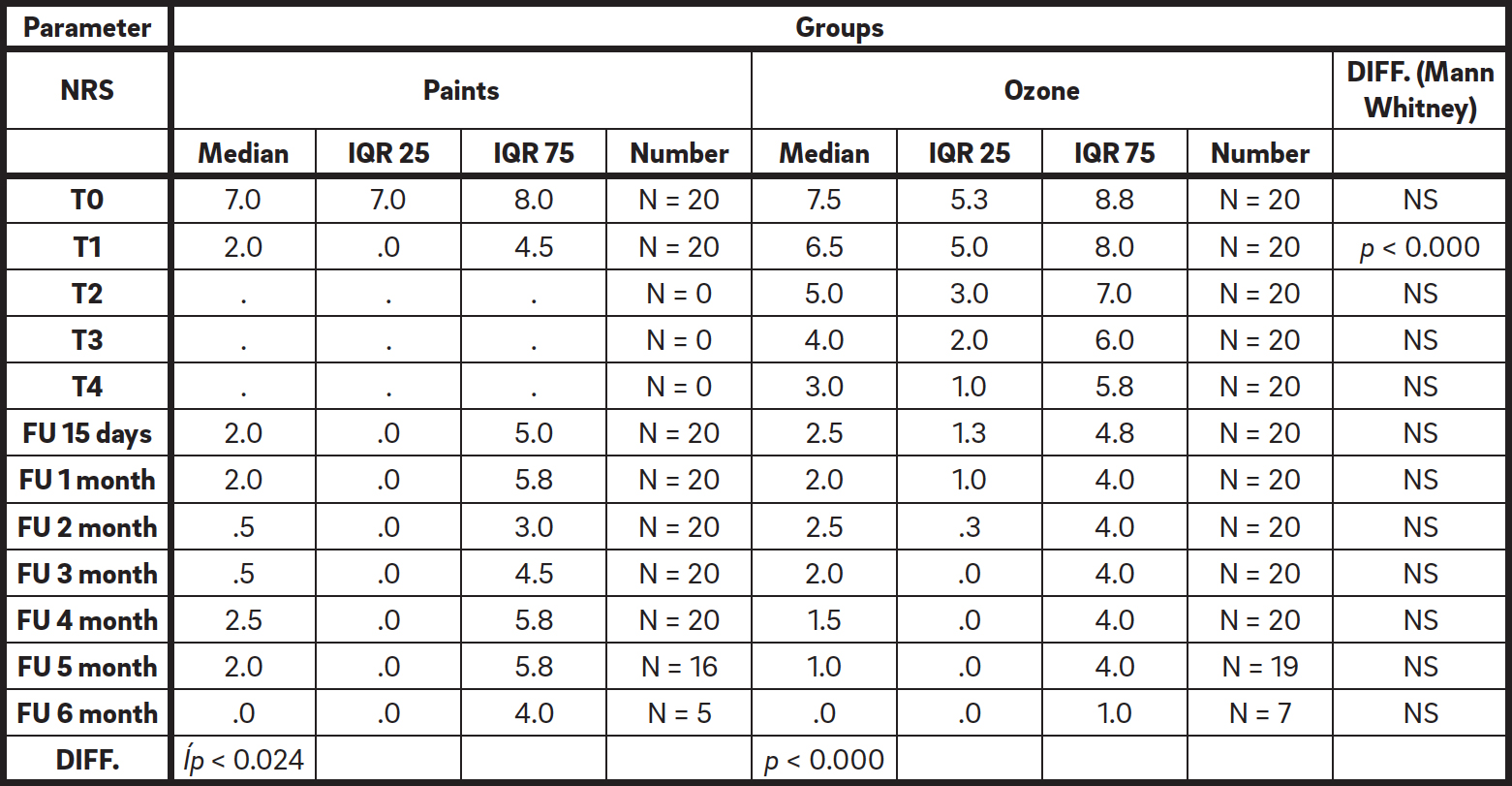
The severity of the dentinal hypersensitivity reported by patients was evaluated according the NRS at the following times: T0, T1, T2, T3, T4 and FU1, FU2, FU3, FU4, FU5, FU6, FU7. Ozone group patients were seen once a week for four consecutive weeks (T1, T2, T3, T4) and later for controls at FU1, FU2, FU3, FU4, FU5, FU6, and FU7. At T0, the NRS in the ozone group was 7.0 (7.0–8.0) and 7.5 (5.3–8.8) in the paint group. Therefore, both groups started at a high degree of pain and were comparable in dentinal hypersensitivity at baseline. Evaluating the trend of NRS values at the various time points within the individual groups using the Friedman test, a gradual improvement of the symptoms was registered in the paint group (p < 0.024) and in the ozone group (p < 0.000). Considering the variation in NRS values within groups before and after the specific treatment, the following results were registered, as reported in Table 1.
Table 1
Trend of NRS values within the two groups at the various time points.
In the paint group, a significant reduction in NRS values was observed (p < 0.000 between T0 and T1) before treatment (T0) and immediately after the application (T1). A significant reduction in NRS values was not observed prior to treatment and at the end of the follow-up for all of the patients (p < 0.003 between T0 and FU6).
In the ozone group, a significant reduction in NRS values was observed before treatment and after the fourth session of ozone therapy (p < 0.000 between T0 and T4). A significant reduction in NRS values was registered prior to treatment and at the end of follow-up for all of the patients (p < 0.000 between T0 and FU6). Concerning the six-month follow-up, a significant difference in NRS values (p < 0.05 between T0 and FU7) was observed.
A significant decrease in NRS values was registered (p < 0.000 according to the Mann– Whitney test) in the paint group. The raw data revealed a progressive decrease in NRS values in patients treated with ozone. Moreover, the follow-ups revealed a further maintenance of the results regarding NRS values at six months.
Discussion
The present study takes into consideration the possibility of extending the use of ozone therapy in the dental field to the treatment of dentinal hypersensitivity. The purpose of the research was to evaluate the effectiveness of ozone in the reduction of pain due to hypersensitivity. From the results obtained, the desensitizing treatment with paint was proven effective in terms of immediate results, attributable to the paint’s ability to form a protective layer, a mechanical barrier on the affected surface. The provisional barrier seals the dentinal tubules, preventing the flow of the dentinal fluid and therefore interrupting nerve stimulation and the consequent perception of pain. The six-month follow-up, however, did not find significant maintenance of the results. This may be due to the inability of adhesive agents to withstand the stress of the oral environment and their undergoing a progressive dissolution that causes the restoration of the initial sensitivity values, as observed by Jain et al.17Jain P, Reinhardt JW, Krell KV. Effect of dentin desensitizers and dentin bonding agents on dentin permeability.
→ Am J Dent. 2000 Feb;13(1):21–7.
In the present study, no significant difference was reported after six months from the first application, but this was probably due to the insufficient sample sizes of patients. Therefore, the use of paint-on desensitizers, whose effectiveness is well established and well documented in the literature, may not be a definitive treatment and may require repeated applications throughout the patient’s life. Duke et al. observed that the action of paint can still be effective up to five months.18Duke ES, Platt JA, Moore KB. Prospective placebo controlled clinical trial of a dentin desensitizing agent. 6 month report.
→ Paper presented at: IADR/AADR/CADR 85th General Session and Exhibition. 2007 Mar 21–24; New Orleans, LA.
In the ozone group, a progressive reduction in NRS values was observed; this can be attributed to the beneficial action of the ozone itself on the tooth surface. In contrast to what was observed by Azarpazooh et al.,19Azarpazooh A, Limeback H. The application of ozone in dentistry: a systematic review of literature.
→ J Dent. 2008 Feb;36(2):104–16. one application of ozone was not sufficient to resolve the pain symptoms linked to dentinal hypersensitivity; the results appeared more evident from the second application on, instead. As observed by Bocci et al., the increase in patency of the tubules caused by the application of ozone on the dental surface may facilitate the entry of minerals in the saliva and desensitizing agents.20Bocci V, Bianchi L, Larini A. The ozone enigma in medicine. The biochemical relationship between ozone and body fluids may account for its biological, therapeutic and toxic effects.
→ Riv Ital Ossigeno-Ozonoterapia. 2003 Oct;2(2):113–20. Italian. In this way, the sealing of the dentinal tubules would be continuous owing to the mineral salts contained in the saliva (mainly calcium and fluoride). According to this theory, it can be assumed that the action of ozone causes a sort of repair of the dentin by modifying its internal structure and therefore leading to longterm effects. The long-term maintenance of the beneficial effect of ozone therapy, as well as the reduction of pain, seems more evident if compared with the use of paint, despite the small numeric sample. Considering that ozone does not occlude the dentinal tubules, its action could be enhanced by the concomitant use of fluoridebased products or amorphous calcium phosphate to create a smear plug in the tubules. In fact, as noted by another study,21Raafat Abdelaziz R, Mosallam RS, Yousry MM. Tubular occlusion of simulated hypersensitive dentin by the combined use of ozone and desensitizing agents.
→ Acta Odontol Scand. 2011 Nov;69(6):395–400. there is a synergy between ozone and fluoride that does not occur with other desensitizers, such as oxalates. For this reason, ozone therapy could be associated with fluoride products in both professional application and at-home care.
Conclusion
Both therapies proved to be effective. The paint demonstrated an immediate desensitizing action, which was not observed in the treatment with ozone. However, in the longer term, the paint did not ensure a significant reduction in NRS values. Treatment with ozone proved to be more effective in the maintenance of long-term results. It can therefore be said that ozone therapy in the management of dentinal hypersensitivity represents an innovative and effective technique, with good maintenance of long-term results.
Acknowledgments
The authors would like to acknowledge Dr. Margherita Cimadori for her fundamental contribution.
Competing interests
None of the authors have any conflict of interest to disclose.
Why did you conduct the research reported on in this paper?
For what reasons could others cite your paper?
How could your study’s findings have an impact on dentistry? What is the relevance of your study’s findings to the daily practice of a dentist?
What are your recommendations for further investigation of the topic of your article?
References
| 1, 6. | ↑ | Canadian Advisory Board on Dentin Hypersensitivity. Consensus-based recommendations for the diagnosis and management of dentin hypersensitivity. → J Can Dent Assoc. 2003 Apr;69(4):221–6. |
| 2. | ↑ | Aranha AC, Pimenta LA, Marchi GM. Clinical evaluation of desensitizing treatments for cervical dentin hypersensitivity. → Braz Oral Res. 2009 Jul-Sep;23(3):333–9. |
| 3, 8, 10. | ↑ | Abbinate A, Ardizzone VC. Cap 23 Trattamento della sensibilità dentinale. → In: Abbinate A, Ardizzone VC. Igienista orale. Teoria e pratica professionale. Milano: Edra editor. 2013 p. 437–8. |
| 4. | ↑ | Miglani S, Aggarwal V, Ahuja B. Dentin hypersensitivity: recent trends in management. → J Conserv Dent. 2010 Oct;13(4);218–24. |
| 5. | ↑ | Brännström M, Astroem A. A study on the mechanism of pain elicited from the dentin. → J Dent Res. 1964 Jul-Aug;43:619–25. |
| 7, 9. | ↑ | Gaffar A. Treating hypersensitivity with fluoride varnish. → Compend Contin Educ Dent. 1999;20(1 Suppl):27–33; quiz 35. |
| 11, 13. | ↑ | Sunnen GV. Ozone in medicine: overview and future directions. → J Adv Med. 1988 Fall;1(3):159–74. |
| 12. | ↑ | Miglani S, Aggarwal V, Ahuja B. Dentin hypersensitivity: recent trends in management. → J Conserv Dent. 2010 Oct;13(4);218–24 |
| 14. | ↑ | Summerfelt ST, Hochheimer JN. Review of ozone processes and applications as an oxidizing agent in aquaculture. → Prog Fish Cult. 1997;59(2):94–105. |
| 15. | ↑ | Lynch E. Ozone: the revolution in dentistry. → Copenhagen: Quintessence. 2004. 300 p. |
| 16, 20. | ↑ | Bocci V, Bianchi L, Larini A. The ozone enigma in medicine. The biochemical relationship between ozone and body fluids may account for its biological, therapeutic and toxic effects. → Riv Ital Ossigeno-Ozonoterapia. 2003 Oct;2(2):113–20. Italian. |
| 17. | ↑ | Jain P, Reinhardt JW, Krell KV. Effect of dentin desensitizers and dentin bonding agents on dentin permeability. → Am J Dent. 2000 Feb;13(1):21–7. |
| 18. | ↑ | Duke ES, Platt JA, Moore KB. Prospective placebo controlled clinical trial of a dentin desensitizing agent. 6 month report. → Paper presented at: IADR/AADR/CADR 85th General Session and Exhibition. 2007 Mar 21–24; New Orleans, LA. |
| 19. | ↑ | Azarpazooh A, Limeback H. The application of ozone in dentistry: a systematic review of literature. → J Dent. 2008 Feb;36(2):104–16. |
| 21. | ↑ | Raafat Abdelaziz R, Mosallam RS, Yousry MM. Tubular occlusion of simulated hypersensitive dentin by the combined use of ozone and desensitizing agents. → Acta Odontol Scand. 2011 Nov;69(6):395–400. |






Leave a Reply
Be the First to Comment!