Immunohistochemical osteopontin expression in bone xenograft in clinical series of maxillary sinus lift
October 2, 2015 / Categories: Digital Dentistry, Implant Dentistry

Galindo Moreno, Pablo

Hernández Cortés, Pedro

Padial Molina, Miguel

Vizoso, María Luisa

Crespo Lora, Vicente

O’Valle Ravassa, Francisco
The objectives of this study were to examine osteopontin (OPN) expression in bone and anorganic bovine bone (ABB) in maxillary sinus grafts after six months of healing and to study its relationship to morphological and immunohistochemical results and to patient variables and habits.
Introduction
Osteopontin (OPN) human gene contains seven exons, spans ~11.1 kb, and maps to the long arm of chromosome 4 (4q13).1Young MF, Kerr JM, Termine JD, Wewer UM, Wang MG, McBride OW, Fisher LW. cDNA cloning, mRNA distribution and heterogeneity, chromosomal location, and RFLP analysis of human osteopontin (OPN). → Genomics. 1990 Aug;7(4):491–502.3Hijiya N, Setoguchi M, Matsuura K, Higuchi Y, Akizuki S, Yamamoto S. Cloning and characterization of the human osteopontin gene and its promoter. → Biochem J. 1994 Oct;303(Pt 1):255–62. OPN is expressed by a single-copy gene as a ~34 kDa2Oldberg A, Franzén A, Heinegård D. Cloning and sequence analysis of rat bone sialoprotein (osteopontin) cDNA reveals an Arg-Gly-Asp cell-binding sequence. → Proc Natl Acad Sci USA. 1986 Dec;83(23):8819–23. nascent protein that is extensively modified by posttranslational events; it is secreted as a noncollagenous acidic bone matrix of single-chain phosphoglycoproteins with diverse functions, including cell-binding activity4Milam SB, Haskin C, Zardeneta G, Chen D, Magnuson VL, Klebe RJ. Cell adhesion proteins in oral biology. → Crit Rev Oral Biol Med. 1991;2(4):451–91. and angiogenesis.5Matusan-Ilijas K, Behrem S, Jonjic N, Zarkovic K, Lucin K. Osteopontin expression correlates with angiogenesis and survival in malignant astrocytoma. → Pathol Oncol Res. 2008 Sep;14(3):293–8. OPN has calcium-binding properties and is expressed by cells in a wide variety of tissues, including bone, tooth and cartilage, and in activated macrophages and lymphocytes.6Sodek J, Ganss B, McKee MD. Osteopontin. → Crit Rev Oral Biol Med. 2000;11(3):279–303.
Data are available on the structure, location and properties of OPN, but the biological function of this protein in bone remains uncertain. OPN influences bone homeostasis by different mechanisms. This polypeptide chain undergoes extensive post-translational modifications, including glycosylation, phosphorylation and sulfation, and the precise modification pattern depends on the species and tissues in which the protein is synthesized.7Taller A, Grohe B, Rogers KA, Goldberg HA, Hunter GK. Specific adsorption of osteopontin and synthetic polypeptides to calcium oxalate monohydrate crystals. → Biophys J. 2007 Sept;93(5):1768–77. The functional significance of post-translational modifications in OPN is poorly understood.
Bone remodeling is a regulated process in which removal via osteoclasts is followed by bone formation via osteoblasts.8Sims NA, Gooi JH. Bone remodeling: multiple cellular interactions required for coupling of bone formation and resorption. → Semin Cell Dev Biol. 2008 Oct;19(5):444–51. The presence of OPN has traditionally been interpreted as an indicator of bone formation. In bone, OPN is produced by osteoblastic cells at various stages of differentiation,9Zohar R, Lee W, Arora P, Cheifetz S, McCulloch C, Sodek J. Single cell analysis of intracellular osteopontin in osteogenic cultures of fetal rat calvarial cells. → J Cell Physiol. 1997 Jan;170(1):88–100. including differentiated osteo – blasts, and by osteocytes.10McKee MD, Nanci A. Osteopontin and the bone remodeling sequence: colloidal-gold immunocytochemistry of an interfacial extracellular matrix protein. → Ann N Y Acad Sci. 1995 Aug;760:177–89.11Sodek J, Chen J, Nagata T, Kasugai S, Todescan R Jr, Li IW, Kim RH. Regulation of osteopontin expression in osteoblasts. → Ann N Y Acad Sci. 1995 Aug;760:223–41. The protein is primarily made by cells of osteoblastic lineage, and it is also expressed by fibroblastic cells in embryonic stroma12Giachelli CM, Schwartz SM, Liaw L. Molecular and cellular biology of osteopontin: potential role in cardiovascular disease. → Trends Cardiovasc Med. 1995 May-Jun;5(3):88–95. and at wound-healing sites.13Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. OPN is found in situ in osteoblasts and accumulates in mineralized bone matrix during endochondral and intramembranous ossification.14Choi ST, Kim JH, Kang EJ, Lee SW, Park MC, Park YB, Lee SK. Osteopontin might be involved in bone remodelling rather than in inflammation in ankylosing spondylitis. → Rheumatology (Oxford). 2008 Dec;47(12):1775–9. It has also been reported to enhance osteoblastic differentiation and proliferation and increase alkaline phosphatase activity.15Jang JH, Kim JH. Improved cellular response of osteoblast cells using recombinant human osteopontin protein produced by Escherichia coli. → Biotechnol Lett 2005 Nov;27(22):1767–70. Increased OPN expression at injury or infection sites likely results from the release of growth factors (e.g., platelet-derived growth factor) or cytokines (e.g., interleukin1) that activate different transcription factors, such as Fos and Jun, which are capable of upregulating OPN transcription.16Denhardt DT, Noda M. Osteopontin expression and function: role in bone remodeling. → J Cell Biochem Suppl. 1998;30–31 Suppl:92–102.
Hence, besides promoting bone formation, OPN has been implicated in bone resorption.17Ishijima M, Ezura Y, Tsuji K, Rittling SR, Kurosawa H, Denhardt DT, Emi M, Nifujia A, Noda M. Osteopontin is associated with nuclear factor κB gene expression during tail-suspension-induced bone loss. → Exp Cell Res. 2006 Oct;312(16):3075–83. Various mechanisms have been proposed to underlie this biological function. Phosphorylation of OPN appears necessary for the inhibition of biological crystal formation and for the formation of calcium carbonate crystals.18Taller A, Grohe B, Rogers KA, Goldberg HA, Hunter GK. Specific adsorption of osteopontin and synthetic polypeptides to calcium oxalate monohydrate crystals. → Biophys J. 2007 Sept;93(5):1768–77. OPN is a potent inhibitor of the mineralization process, because it is binding to hydroxyapatite (HA), inhibits the formation of HA crystals19Goldberg HA, Warner KJ, Li MC, Hunter GK. Binding of bone sialoprotein, osteopontin and synthetic polypeptides to hydroxyapatite. → Connect Tissue Res. 2001;42(1):25–37. and the growth of HA crystals,20Pampena DA, Robertson KA, Litvinova O, Lajoie G, Goldberg HA, Hunter GK. Inhibition of hydroxyapatite formation by osteopontin phosphopeptides. → Biochem J. 2004 Mar;378(3):1083–7. and promotes the inhibition of bone mineralization.21Altıntaş A, Saruhan-Direskeneli G, Benbir G, Demir M, Purisa S. The role of osteopontin: a shared pathway in the pathogenesis of multiple sclerosis and osteoporosis? → J Neurol Sci. 2009 Jan;276(1–2):41–4. OPN plays an important role in osteoclastogenesis and osteoclast activity. Its expression is upregulated during the maturation of monocytes into macrophages,22Krause SW, Rehli M, Kreutz M, Schwarzfischer L, Paulauskis JD, Andreesen R. Differential screening identifies genetic markers of monocyte to macrophage maturation. → J Leukoc Biol. 1996 Oct;60(4):540–5. a process that presumably occurs as circulating monocytes extravasate and migrate through the tissue.23Denhardt DT, Noda M. Osteopontin expression and function: role in bone remodeling. → J Cell Biochem Suppl. 1998;30–31 Suppl:92–102. Parathyroid hormone- induced RANKL signaling normally augments the number and activation of osteoclasts, but this increase is disrupted in the absence of OPN.24Ihara H, Denhardt DT, Furuya K, Yamashita T, Muguruma Y, Tsuji K, Hruska KA, Higashio K, Enomoto S, Nifuji A, Rittling SR, Noda M. Parathyroid hormoneinduced bone resorption does not occur in the absence of osteopontin. → J Biol Chem. 2001 Apr;276(16):13065–71. The neutralization of OPN suppresses osteoclastogenesis in vitro, whereas its addition enhances osteoclastogenesis in OPN-/- cells.25Standal T, Borset M, Sundan A. Role of osteopontin in adhesion, migration, cell survival and bone remodeling. → Exp Oncol. 2004 Sep;26(3):179–84. However, Chellaiah et al. reported an increase in the number of osteoclasts in OPN-/- mice as a compensatory mechanism for the decreased activity of OPN-/- osteoclasts, because OPN-deficient osteoclasts do not migrate and are unable to resorb bone.26Chellaiah MA, Kizer N, Biswas R, Alvarez U, Strauss-Schoenberger J, Rifas L, Rittling SR, Denhardt DT, Hruska KA. Osteopontin deficiency produces osteoclast dysfunction due to reduced CD44 surface expression. → Mol Biol Cell. 2003 Jan;14(1):173–89.27Tani-Ishii N, Tsunoda A, Umemoto T. Osteopontin antisense deoxyoligonucleotides inhibit bone resorption by mouse osteoclasts in vitro. → J Periodontal Res. 1997 Aug;32(6):480–6. Hence, bone-resorbing activity could only partially be restored by exogenous OPN,28Chellaiah MA, Kizer N, Biswas R, Alvarez U, Strauss-Schoenberger J, Rifas L, Rittling SR, Denhardt DT, Hruska KA. Osteopontin deficiency produces osteoclast dysfunction due to reduced CD44 surface expression. → Mol Biol Cell. 2003 Jan;14(1):173–89. indicating that autocrine OPN is important for osteoclast activity.29Suzuki K, Zhu B, Rittling SR, Denhardt DT, Goldberg HA, McCulloch CA, Sodek J. Colocalization of intracellular osteopontin with CD44 is associated with migration, cell fusion, and resorption in osteoclasts. → J Bone Miner Res. 2002 Aug; 17(8):1486–97.Anorganic bovine bone (ABB) is a deproteinized, sterilized bovine cancellous bone comprising calcium-deficient carbonate apatite.30Carinci F, Piattelli A, Degidi M, Palmieri A, Perrotti V, Scapoli L, Martinelli M, Laino G, Pezzetti F. Genetic effects of anorganic bovine bone (Bio-Oss) on osteoblast-like MG63 cells. → Arch Oral Biol. 2006 Feb;51(2):154–63. ABB is frequently utilized as a bone substitute in maxillary sinus lift procedures when insufficient autogenous cortical bone (ACB) is available for the graft.31Tadjoedin ES, De Lange GL, Bronckers AL, Lyaruu DM, Burger EH. Deproteinized cancellous bovine bone (Bio-Oss) as bone substitute for sinus floor elevation: a retrospective, histomorphometrical study of five cases. → J Clin Periodontol. 2003 Mar;30(3):261–70. ABB particles are similar to human cancellous bone in crystalline and morphological structure.32Yildirim M, Spiekermann H, Biesterfeld S, Edelhoff D. Maxillary sinus augmentation using xenogenic bone substitute material Bio-Oss in combination with venous blood: a histologic and histomorphometric study in humans. → Clin Oral Implants Res. 2000 Jun;11(3):217–29. They are natural, osteoconductive bone substitutes that promote bone growth in periodontal and maxillofacial osseous defects. The particles provide a scaffold and a matrix for bone cell migration and are integrated into the natural physiological remodeling process. It has been suggested that deproteinized cancellous bovine bone can induce new bone formation through osteoinductive mechanisms.33Schwartz Z, Weesner T, Van Dijk S, Cochran DL, Mellonig JT, Lohmann CH, Carnes DL, Goldstein M, Dean DD, Boyan BD. Ability of deproteinized cancellous bovine bone to induce new bone formation. → J Periodontol. 2000 Aug;71(8):1258–69. It has also been reported that the application of ABB in a collagenous matrix induces the formation of membranous and endochondral bone in vivo and that ABB exerts high angiogenic activity.34Galindo-Moreno P, Padial-Molina M, Fernández-Barbero JE, Mesa F, Rodríguez- Martínez D, O’Valle F. Optimal microvessel density from composite graft of autogenous maxillary cortical bone and anorganic bovine bone in sinus augmentation: influence of clinical variables. → Clin Oral Implants Res. 2010 Feb;21(2):221–7. In previous studies, however, no OPN was detected in bovine bone slices, and no staining was observed in osteocytes, blood vessels, cement lines or typical sites of OPN expression.35Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E, Gowen M. Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. → J Bone Miner Res. 1995 Nov;10(11):1666–80. In an animal study, Araújo et al. described OPN expression in ABB particles during early healing of the post-extraction socket.36Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. Our group described a similar phenomenon in humans during late healing after sinus grafting, observing OPN expression not only in the ABB particles, but also within their canalicular system. These observations differ from previous findings in ultrastructural studies in a rat model that suggested that OPN accumulated at the mineral front and was progressively incorporated deeper into the bone, but by the further deposition of new bone matrix.37McKee MD, Farach-Carson MC, Butler WT, Hauschka PV, Nanci A. Ultrastructural immunolocalization of noncollagenous (osteopontin and osteocalcin) and plasma (albumin and α2HS-glycoprotein) proteins in rat bone. → J Bone Miner Res. 1993 Apr;8(4):485–96.
The objectives of this study were to examine OPN expression in bone and ABB in maxillary sinus grafts after six months of healing and to study its relationship to morphological and immunohistochemical results and to patient variables and habits.
Materials & methods
Study design and subject recruitment
This clinical case series was reviewed and approved by the institutional review board of the University of Granada Faculty of Dentistry (Spain) prior to subject recruitment. The study was conducted according to the principles of the Declaration of Helsinki for experimentation with human subjects.38Schücklenk U, Ashcroft R. International research ethics. → Bioethics. 2000 Apr;14(2):158–72.
Totally or partially edentulous patients needing a sinus lift were screened and included in the study if they met the following inclusion criteria: age between 18 and 85 years, Physical Status I or II according to the American Society of Anesthesiologists,39American Society of Anesthesiologists. ASA Physical Status Classification System [Internet]. → Schaumburg, Ill.: American Society of Anesthesiologists; [updated 2014 Oct 15; cited 2015 Jul 1]. Available from: http://www.asahq.org/resources/clinicalinformation/ asa-physical-status-classifica tion-system. absence of uncontrolled systemic disease or a condition known to alter bone metabolism (e.g., osteoporosis or diabetes mellitus), O’Leary plaque score of ≤ 20%,40O’Leary TJ, Drake RB, Naylor JE. The plaque control record. → J Periodontol. 1972 Jan;43(1):38. and ≤ 5 mm of remaining bone height from measurement on a panoramic radiograph.41Wang HL, Katranji A. ABC sinus augmentation classification. → Int J Periodontics Restorative Dent. 2008 Jul-Aug;28(4):383–9.42Zinner ID, Small SA. Sinus-lift graft: using the maxillary sinuses to support implants. → J Am Dent Assoc. 1996 Jan;127(1):51–7. Exclusion criteria were the following: antibiotic intake in the previous three months, prescription for more than six months of medications known to modify bone metabolism (e.g., bisphosphonates or corticosteroids), pregnancy or intention to become pregnant at the time of the screening, the presence of an untreated chronic sinus condition (e.g., cyst or tumor) or sepsis, a history of cancer or radiation to the oral cavity, complications of these conditions affecting the sinus area, and consumption of > 10 cigarettes/day. Patients smoking up to 10 cigarettes/day43Levin L, Herzberg R, Dolev E, Schwartz- Arad D. Smoking and complications of onlay bone grafts and sinus lift operations. → Int J Oral Maxillofac Implants. 2004 May-Jun;19(3):369–73. and alcohol consumers were included in the study. For the statistical analysis, patients who smoked ≥ 1 cigarettes/day were considered smokers and those having ≥ 1 alcohol- containing drinks/day (> 10 g of alcohol/day)44Galindo-Moreno P, Fauri M, Ávila-Ortiz G, Fernández-Barbero JE, Cabrera-León A, Sánchez-Fernández E. Influence of alcohol and tobacco habits on peri-implant marginal bone loss: a prospective study. → Clin Oral Implants Res. 2005 Oct;16(5):579–86. were considered alcohol consumers. Patients who met the inclusion and exclusion criteria were required to read, understand and sign the informed consent form before being enrolled in the study.
Surgical procedures
Patients were asked to take 875/125 mg amoxicillin/ clavulanate (or, if allergic to penicillin, 300 mg clindamycin) t.i.d. for ten days, starting two days before the surgery to minimize infection risk. All surgical procedures were performed under local anesthesia (articaine with epinephrine 40/0.01 mg/ml, Sanofi-Aventis Deutschland, Frankfurt/Main, Germany). The procedure proposed by Galindo-Moreno et al.45Galindo-Moreno P, Ávila G, Fernández- Barbero JE, Aguilar M, Sánchez-Fernández E, Cutando A, Wang HL. Evaluation of sinus floor elevation using a composite bone graft mixture. → Clin Oral Implants Res. 2007 Jun;18(3):376–82. was followed, using a bone scraper (Safescraper, Meta, Reggio Emilia, Italy) to harvest ACB from the lateral wall and expose the Schneiderian membrane. After the membrane lift, sinus cavities were grafted with scraped ACB in combination with ABB particles sized between 250 and 1,000 μm (Geistlich Bio-Oss, Geistlich Pharma, Wolhusen, Switzerland); the ratio of ACB to ABB in the composite graft was 1:1 v/v.46Galindo-Moreno P, Ávila G, Fernández- Barbero JE, Mesa F, O’Valle-Ravassa F, Wang HL. Clinical and histologic comparison of two different composite grafts for sinus augmentation: a pilot clinical trial. → Clin Oral Implants Res. 2008 Aug;19(8):755–9.A maximum of 5 cc of graft material was used per sinus cavity. After bone grafting, an absorbable collagen membrane (Geistlich Bio-Gide, Geistlich Pharma, Wolhusen, Switzerland) was placed over the lateral aspect of the bony window. Flaps were then carefully approximated and sutured with 3-0 surgical silk (Laboratorio Aragó, Barcelona, Spain) by primary intention.
After a six-month healing period, a trephine (internal and external diameters of 3 mm and 4 mm, respectively) was used to harvest bone core biopsies from the alveolar crest in which implants were prosthetically planned. Implants (OsseoSpeed, Astra Tech, Mölndal, Sweden; Microdent, Microdent Implant System, Barcelona, Spain) were placed in a two-stage approach.
Histological study
The trephine biopsies were fixed in 10% buffered formalin for 24 h, decalcified in Decalcifier I (Surgipath Europe, Peterborough, UK), containing formaldehyde (10% w/v), formic acid (8% w/v) and methanol (1% w/v), for 24 h at 37 ºC in an oven and embedded in paraffin. Then, 4 μm sections were cut along the central axis of the biopsies and dewaxed and hydrated for staining with hematoxylin–eosin, periodic acid–Schiff, Masson’s trichrome and Goldner’s trichrome. A millimeter scale in the eyepiece of a BH2 microscope (Olympus Optical, Tokyo, Japan) with a 40× objective was used to count osteoblasts, osteoclasts and osteocytes per mm². Results were expressed in terms of the number of positive cells per mm2.
Bone histomorphometry was performed semi-automatically on Masson’s trichromestained sections, assessing ten randomized images with a 10× objective, using a microscope equipped with a digital camera (DP70, Olympus Optical, Tokyo, Japan) connected to a computer and applying ImageJ software (Version 1.48; developed by the U.S. National Institutes of Health, Bethesda, Md.). Separate quantifications of vital bone, ABB particles and connective tissue were performed, expressing the results as percentages of each compartment.
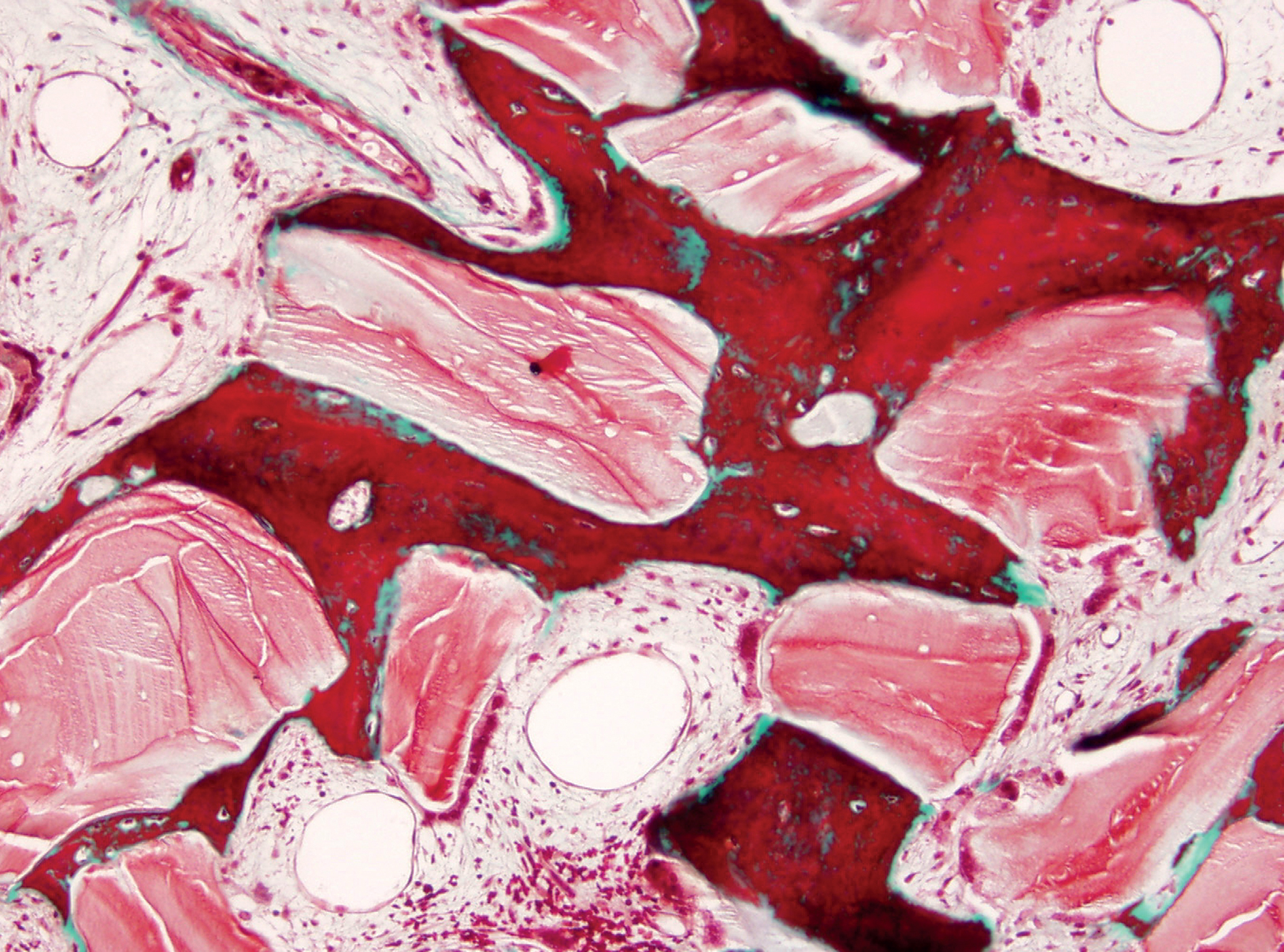
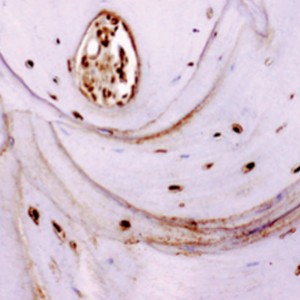
Fig. 1
Trabecular bone formed throughout the ABB particles in a maxillary sinus lift biopsy (Masson’s trichrome stain, 10×).
Immunohistochemical analysis
Decalcified and paraffin-embedded sections were dewaxed, hydrated and heat-treated in 1 mM EDTA buffer for antigenic unmasking. Sections were incubated for 60 min at room temperature with pre-diluted OPN polyclonal antibody to identify cellular and interstitial expression and with the following pre-diluted monoclonal antibodies (all from Master Diagnóstica, Granada, Spain): CD34 (clone QBEnd/10) to identify endothelial cells; CD56 (clone 56C04/123A8) to identify osteoblasts; tartrate-resistant acid phosphatase (TRAP; clone 26E5) to identify osteoclasts; CD68 (clone KP1) to identify monocytes and macrophages; and vimentin (clone V9) to identify mesenchymal cells (as positive control). The immunohistochemical study was done on an automatic immunostainer (Autostainer 480S, LabVision, Fremont, Calif., U.S.), using the micropolymer-peroxidase-based method followed by development with diaminobenzidine (Ultravision Quanto, Master Diagnóstica, Granada, Spain). A millimeter scale in the eyepiece of a BH2 microscope (Olympus Optical, Tokyo, Japan) with a 40× objective was used to count the number of positive cells and vessels per mm².
Statistical analysis
The Shapiro–Wilk test was used to assess the normality of variables. After descriptive analysis, the Student’s t-test or Welch test for unequal variances and the Spearman correlation coefficient (rho) were used to evaluate the significance of differences. Clinical, morphological and morphometric variables were compared between the presence and absence of OPN in cells and ABB particles using the Student’s t-test. A p < 0.05 was considered significant. IBM SPSS Statistics for Windows (Version 20.0; IBM, Armonk, N.Y., U.S.) was used for the data analyses.
Results
Histological and histomorphometric results
After six months, a normal woven and lamellar pattern of trabecular bone had formed throug – hout the graft in all patients who had received ABB plus ACB (1:1) grafts, and biopsies from the augmentation area contained this trabecular bone in different proportions. Image analysis revealed 32.75 ± 14.0% vital bone, 39.49 ± 17.4% connective tissue, and 27.75 ± 21.8% remnant ABB particles (Fig. 1). ABB particles were detectable in the trabecular bone in a slightly smaller proportion than in the original graft. In the pristine bone, 48.93 ± 15.4% was vital bone and 51.07 ± 20.8% connective tissue.
Immunohistochemical results
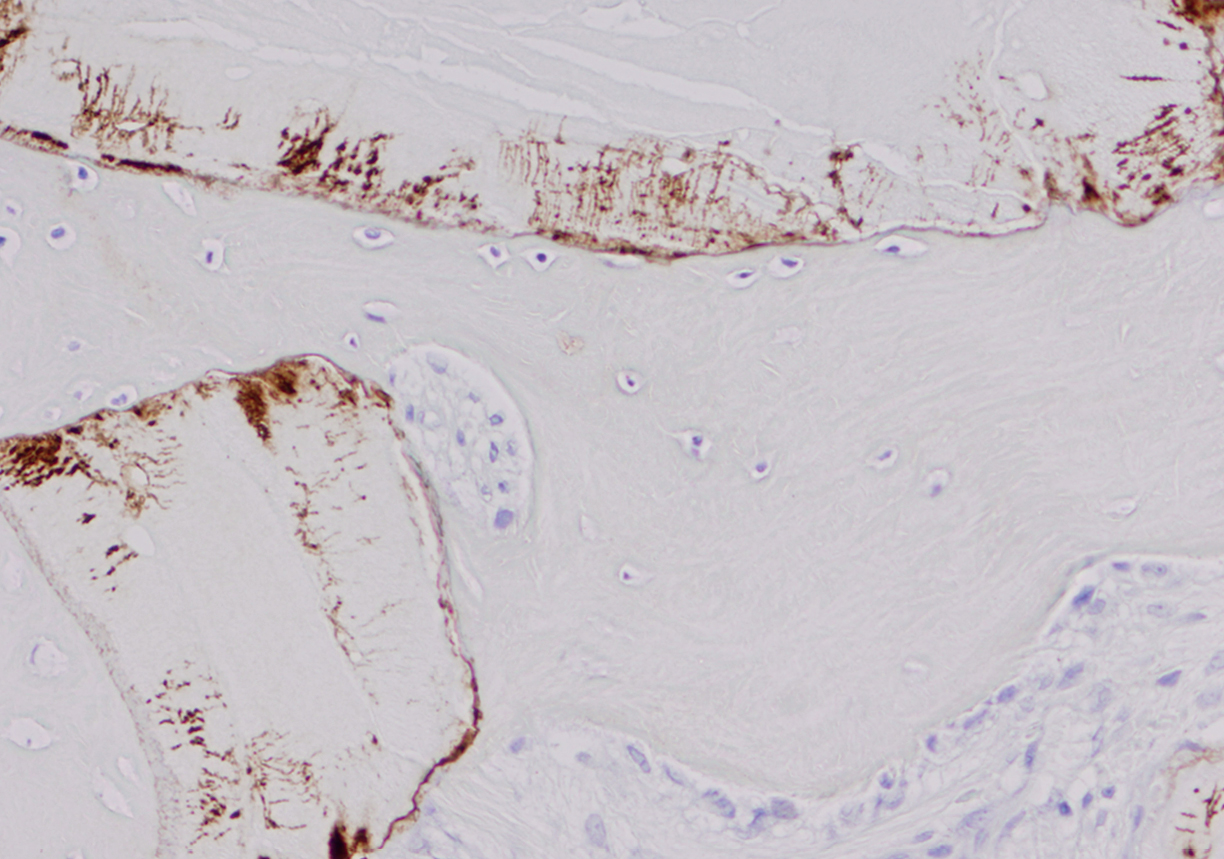
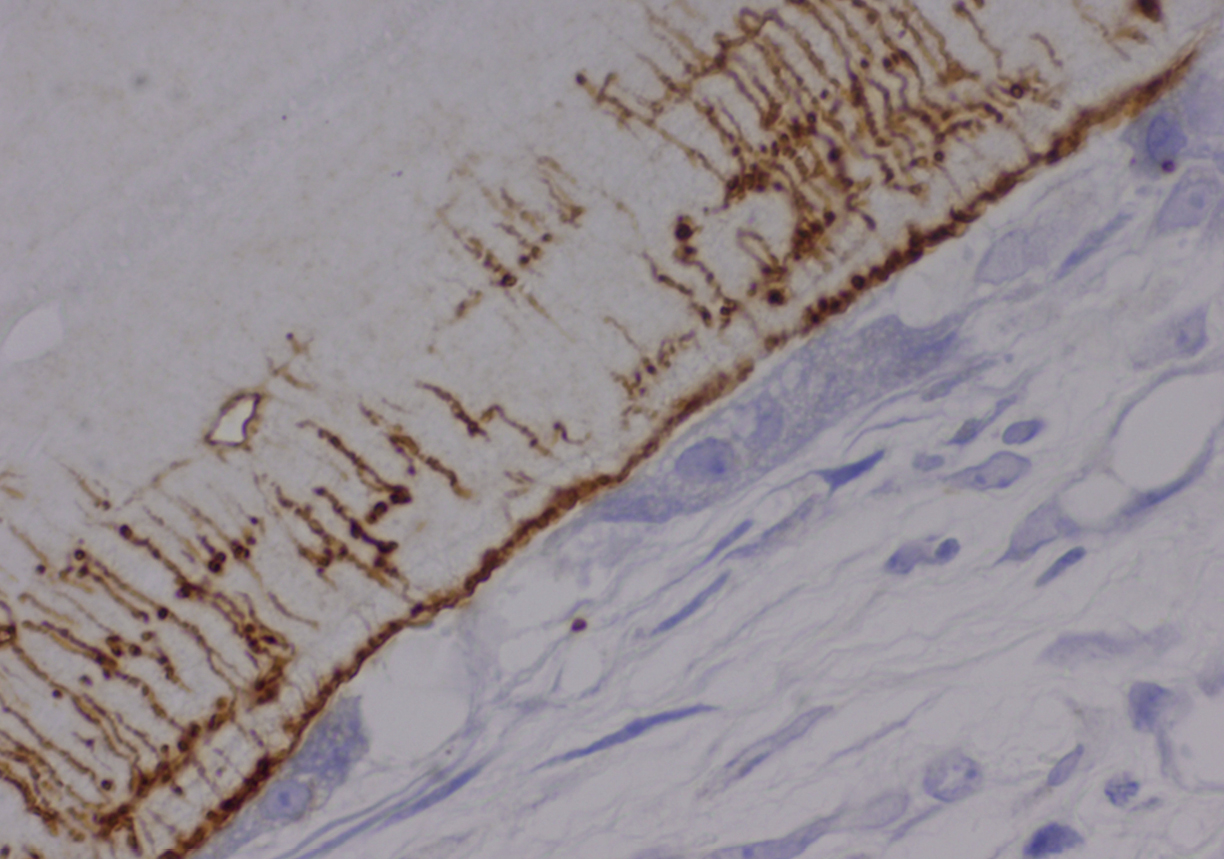
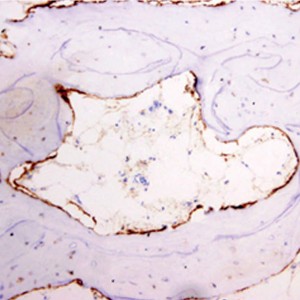
OPN expression was diffuse in 77.5% (31/40) of ABB samples and focal in 22.5% (9/40); it was diffuse in 80% (8/10) of pristine bone samples and focal in 20% (2/10). The presence of OPN immunostaining on ABB particles was intense in 45% of maxillary sinus lift biopsies, moderate in 27.5%, mild in 10%, and absent in 17.5%; it was distributed within the lacuno-canalicular system of ABB particles and on their surface close to osteoclast-like cells (Fig. 2).
Immunohistochemical expression of OPN.
(a) OPN location on boundary of ABB particles with trabecular bone (micropolymer-peroxidasebased method, 20×).
(b) Detail of OPN deposits in lacuno-canalicular network and location on boundary of ABB particles near to osteoclast (micropolymerperoxidase- based method, 60×).
At six months, OPN expression was principally observed at the interstitial boundary of bone with ABB particles and within lacunae and bone canaliculi, forming a star shape (Fig. 2), with no expression in the trabecular bone or interstitium. Cortical OPN expression was directly correlated with the number of osteocytes per mm2 (rho coefficient = 0.405, p = 0.045, Spearman test), with OPN expression in cement lines (rho coefficient = 0.757, p < 0.001, Spearman test) and with OPN expression in osteocytes (rho coefficient = 0.432, p = 0.012, Spearman test).
A direct correlation was found between OPN expression in ABB particles and in monocytes and macrophages (CD68-positive cells; rho coefficient = 0.583, p = 0.009, Spearman test). OPN expression in osteocytes was inversely correlated with the number of osteoblasts (CD56-positive cells) per mm2 (rho coefficient = -0.828, p = 0.042, Spearman test).
A vascular bed was formed in the nonmineralized tissue by vessels of different calibers in the graft area and by capillary vessels among the adipocytes in the bone marrow area, with a mean in the biopsies of 86.28 ± 56.6 CD34- positive vessels per mm2. OPN expression in osteocytes was directly correlated with the number of vessels per mm2 (rho coefficient = 0.828, p = 0.042, Spearman test).
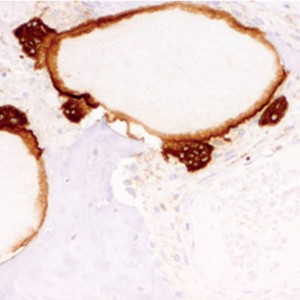
TRAP expression was directly correlated with the count per mm2 of osteoclasts (rho coefficient = 0.532, p = 0.015, Spearman test), monocytes and macrophages (rho coefficient = 0.622, p = 0.008, Spearman test), and osteoblasts (rho coefficient = 0.391, p = 0.048, Spearman test). TRAP expression was correlated with the local and diffuse expression of OPN (rho coefficient = 0.439, p = 0.022, Spearman test; Fig. 3).
Discussion
In this study of bone xenografts in a clinical series of maxillary sinus lift, immunohistochemical OPN expression was detected not only in osteocytes and on ABB particles, but also within the lacuno-canalicular system of ABB particles and close to osteoclast-like cells on their surface.
Bone formation or resorption requires adhesion molecules (arginine–glycine–asparagine sequences), such as fibronectin, fibrinogen, vitronectin, Type I collagen, OPN or bone sialoprotein, to attach osteoblasts or osteoclasts to surfaces for remodeling.47Hallman M, Thor A. Bone substitutes and growth factors as an alternative/complement to autogenous bone for grafting in implant dentistry. → Periodontol 2000. 2008 Jun;47(1):172–92. Because ABB particles are free of proteins,48Wenz B, Oesch B, Horst M. Analysis of the risk of transmitting bovine spongiform encephalopathy through bone grafts derived from bovine bone. → Biomaterials. 2001 Jun;22(12):1599–606. protein expression on the particles must derive from proteins absorbed from the environment. The above chemotactic factors may have stimulated and directed the migration of cells to the foreign material.49Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64.
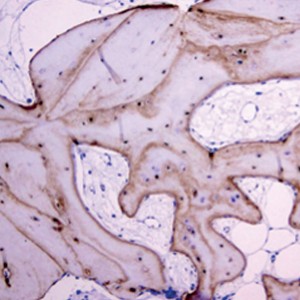
(a) Immunohistochemical expression of OPN in osteocytes and cement lines in pristine bone (micropolymerperoxidase-based method, 10×).
(b) Detail of OPN expression in osteocytes (micropolymerperoxidase-based method, 20×).
(c) Immunohistochemical expression of CD56 in osteoblasts around trabecular bone (micropolymerperoxidase-based method, 10×).
(d) Immunohistochemical expression of TRAP in osteoclasts around ABB particles (micropolymerperoxidase-based method, 40×).
Microchannel pores (< 10 μm) of ABB may be relevant for osteogenic cell attachment, migration, proliferation and differentiation.50Habibovic P, Yuan H, van der Valk CM, Meijer G, van Blitterswijk CA, de Groot K. 3D microenvironment as essential element for osteoinduction by biomaterials. → Biomaterials. 2005 Jun;26(17):3565–75. At the same time, the inner surface of ABB particles becomes considerably enlarged, favoring the formation of new vessels and therefore the inward growth of new bone within the particles.51Galindo-Moreno P, Padial-Molina M, Fernández-Barbero JE, Mesa F, Rodríguez- Martínez D, O’Valle F. Optimal microvessel density from composite graft of autogenous maxillary cortical bone and anorganic bovine bone in sinus augmentation: influence of clinical variables. → Clin Oral Implants Res. 2010 Feb;21(2):221–7. A greater microporosity also expands the scaffold surface and may enhance cytokine adsorption.52Weibrich G, Trettin R, Gnoth SH, Götz H, Duschner H, Wagner W. Bestimmung der Größe der spezifischen Oberfläche von Knochenersatzmaterialien mittels Gasadsorption [Determining the size of the specific surface of bone substitutes with gas adsorption]. → Mund Kiefer Gesichtschir. 2000 May;4(3):148–52. German. The interconnectivity of the pores in ABB particles and their hydrophilic properties explain the presence of OPN within the lacuno-canalicular system. This explanation is supported by the distribution of OPN found in our samples from the surface to the core of the particles, with the lacuno-canalicular system of the remnant ABB particles clearly depicted by the staining. Although the biological relevance of this finding remains unclear, it may be related to the cellular recolonization and revascularization of ABB that has been observed after six months of healing.53Galindo-Moreno P, Padial-Molina M, Fernández-Barbero JE, Mesa F, Rodríguez- Martínez D, O’Valle F. Optimal microvessel density from composite graft of autogenous maxillary cortical bone and anorganic bovine bone in sinus augmentation: influence of clinical variables. → Clin Oral Implants Res. 2010 Feb;21(2):221–7. In this previous study, a direct correlation was found between OPN expression in osteocytes and CD34-positive endothelial cells (rho coefficient = 0.828, p = 0.042, Spearman test). As already noted, one of the functions of OPN is related to angiogenesis,54Matusan-Ilijas K, Behrem S, Jonjic N, Zarkovic K, Lucin K. Osteopontin expression correlates with angiogenesis and survival in malignant astrocytoma. → Pathol Oncol Res. 2008 Sep;14(3):293–8. which is impaired in OPN-deficient mice.55Duvall CL, Taylor WR, Weiss D, Wojtowicz AM, Guldberg RE. Impaired angiogenesis, early callus formation, and late stage remodeling in fracture healing of osteopontin-deficient mice. → J Bone Miner Res. 2007 Feb;22(2):286–97. Images of ABB particles after six months of graft maturation were compatible with neovascularization and central resorption, implying the integration of this biomaterial within the functional and biomechanical system of the neoformed bone.
Whereas most noncollagenous proteins are more or less homogeneously dispersed throughout bone, ultrastructural immunocytochemical studies have consistently found OPN to be predominantly distributed at cement lines in remodeling bone and at laminae limitans.56McKee MD, Nanci A. Osteopontin at mineralized tissue interfaces in bone, teeth, and osseointegrated implants: ultrastructural distribution and implications for mineralized tissue formation, turnover, and repair. Microsc Res Tech. 1996 Feb;33(2):141–64.57McKee MD, Nanci A. Osteopontin: an interfacial extracellular matrix protein in mineralized tissues. → Connect Tissue Res. 1996 Jan;35(1–4):197–205. These sites represent matrix–matrix and cell–matrix boundaries, respectively, and are therefore important in the bone formation process.58Sodek J, Ganss B, McKee MD. Osteopontin. → Crit Rev Oral Biol Med. 2000;11(3):279–303. Cement lines demarcate the boundary between older and newer bone and characteristically have a high OPN content. The origin of OPN in the cement line has been controversial,59Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E, Gowen M. Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. → J Bone Miner Res. 1995 Nov;10(11):1666–80.60McKee MD, Nanci A. Osteopontin deposition in remodeling bone: an osteoblast mediated event. → J Bone Miner Res. 1996 Jun;11(6):873–5. and osteoclasts and osteoblasts may both be involved. OPN may initially play a role in osteoblast adhesion or early calcification events in the cement layer.61Ganss B, Kim RH, Sodek J. Bone sialoprotein. → Crit Rev Oral Biol Med. 1999;10(1):79–98. Subsequently, the more diffuse distribution of OPN throughout the bone matrix may influence osteoclast activity during resorption and the transformation of woven and lamellar bone.62Sodek J, Ganss B, McKee MD. Osteopontin. → Crit Rev Oral Biol Med. 2000;11(3):279–303. Our findings support this proposition, given that OPN expression in the cement lines may indicate bone formation or destruction as a function of the cells attached to the particle surface. However, this dual behavior of OPN remains unclear.
OPN can be expressed in numerous tissues63Kunii Y, Niwa S, Hagiwara Y, Maeda M, Seitoh T, Suzuki T. The immunohistochemical expression profile of osteopontin in normal human tissues using two site-specific antibodies reveals a wide distribution of positive cells and extensive expression in the central and peripheral nervous systems. → Med Mol Morphol. 2009 Sep;42(3):155–61. and by multiple cell types,64Choi ST, Kim JH, Kang EJ, Lee SW, Park MC, Park YB, Lee SK. Osteopontin might be involved in bone remodelling rather than in inflammation in ankylosing spondylitis. → Rheumatology (Oxford). 2008 Dec;47(12):1775–9. including osteoblasts and osteoclasts.65Merry K, Dodds R, Littlewood A, Gowen M. Expression of osteopontin mRNA by osteoclasts and osteoblasts in modelling adult human bone. → J Cell Sci. 1993 Apr;104(4):1013–20. Kunii et al. confirmed the expression of OPN mRNA and protein in human osteocytes66Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E, Gowen M. Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. → J Bone Miner Res. 1995 Nov;10(11):1666–80. using different antibodies.67Kunii Y, Niwa S, Hagiwara Y, Maeda M, Seitoh T, Suzuki T. The immunohistochemical expression profile of osteopontin in normal human tissues using two site-specific antibodies reveals a wide distribution of positive cells and extensive expression in the central and peripheral nervous systems. → Med Mol Morphol. 2009 Sep;42(3):155–61. Osteocytes have multiple functions and regulate different processes in bone homeostasis.68Bonewald L. Osteocytes as multifunctional cells. → J Musculoskelet Neuronal Interact. 2006 Oct-Dec;6(4):331–3. After six months of healing, osteocytes present in the new vital bone in our biopsies consistently expressed OPN. OPN expression in osteocytes was inversely correlated with the count of osteoblasts (CD56- positive cells) per mm2 (rho coefficient = -0.828, p = 0.042, Spearman test). Araújo et al. detected OPN expression on ABB particles in dogs during the early stages of healing.69Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. According to their study, after the migration of polymorphonuclear cells to the ABB surface, these cells are replaced by TRAP-positive cells (osteoclasts), which will remove material from the surface of the xenograft.70Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. Subsequently, osteoblasts will attach to the remodeled surface and bone apposition will begin. However, OPN behavior is likely different during the late stage of healing. In the present samples, osteoblasts were rarely observed in the ABB particles, and a significant inverse relationship was found between OPN expression and CD56 expression (rho coefficient = -828, p = 0.04, Spearman test). This higher expression with fewer osteoblasts and the positive correlation between OPN and CD68 expression (rho coefficient = 0.583, p = 0.009; rho coefficient = 0.938 p = 0.006; Spearman test, respectively) indicate that OPN expression on ABB particles promotes resorption at six months of healing. In support of this hypothesis, it is known that OPN can both activate osteoclasts for bone matrix resorption and induce their migration in an αvβ3 integrin-dependent manner.71Holt I, Marshall MJ. Integrin subunit β3 plays a crucial role in the movement of osteoclasts from the periosteum to the bone surface. → J Cell Physiol. 1998 Apr;175(1):1–9.72Chellaiah MA, Hruska KA. The integrin αvβ3 and CD44 regulate the actions of osteopontin on osteoclast motility. → Calcif Tissue Int. 2003 Mar;72(3):197–205. Nakamura et al. demonstrated that αvβ3 integrin plays a key role in oste oclast migration, which is essential for efficient osteoclastic bone resorption.73Nakamura I, Pilkington MF, Lakkakorpi PT, Lipfert L, Sims SM, Dixon SJ, Rodan GA, Duong LT. Role of alpha(v)beta(3) integrin in osteoclast migration and formation of the sealing zone. → J Cell Sci. 1999 Nov;112(22):3985–93. Perrotti et al. reported that multinucleated cells generated on ABB were positive for the αvβ3 subunit of vitronectin,74Perrotti V, Nicholls BM, Horton MA, Piattelli A. Human osteoclast formation and activity on a xenogenous bone mineral. → J Biomed Mater Res A. 2009 Jun;90A(1):238–46. the main integrin-mediating osteoclast cell attachment to the bone matrix;75Nakamura I, Duong LT, Rodan SB, Rodan GA. Involvement of αvβ3 integrins in osteoclast function. → J Bone Miner Metab. 2007 Nov;25(6):337–44. hence, this mechanism may be functional on ABB particles. OPN is a ligand for the αvβ3 integrin through an RGD sequence. 76Reinholt FP, Hultenby K, Oldberg A, Heinegård D. Osteopontin—a possible anchor of osteoclasts to bone. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4473–5. The osteoclast cell membrane must be sealed to the substrate by means of cell surface receptors and proteins of the integrin family before beginning its proteolytic activity. The binding of OPN to the αvβ3 integrin in the sealing zone or podosomes appears essential to the reorganization of the actin cytoskeleton for osteoclast motility.77Ory S, Brazier H, Pawlak G, Blangy A. Rho GTPases in osteoclasts: orchestrators of podosome arrangement. Eur J Cell Biol. 2008 Sep;87(8-9):469–77. Additionally, osteoclast adhesion and osteoclast migration are mediated by phosphorylated OPN,78Saad FA, Salih E, Glimcher MJ. Identification of osteopontin phosphorylation sites involved in bone remodeling and inhibition of pathological calcification. J Cell Biochem. 2008 Feb;103(3):852–6. and this biological event is regulated by endogenous TRAP.79Ek-Rylander B, Andersson G. Osteoclast migration on phosphorylated osteopontin is regulated by endogenous tartrateresistant acid phosphatase. Exp Cell Res. 2010 Feb;316(3):443–51. Interestingly, TRAP and OPN expressions showed similar patterns on ABB particle surfaces at six months of healing in our samples, indicating the close relationship between OPN expression and the osteoclastic resorption of ABB. Questions have been raised about this resorption of the particles,80Schlegel AK, Donath K. BIO-OSS—a resorbable bone substitute? J Long Term Eff Med Implants. 1998;8(3–4):201–9. References 00_JOSR_A4_Moreno_Layout 1 18.09.15 22:17 Seite 981Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. but this proposition is supported by our histological findings on these particles of bone-remodeling units with multinucleated cells in different stages of differentiation (CD68 positive and TRAP positive) that promote these phenomena. In our view, these results may in part explain OPN-mediated ABB resorption during the late stage of graft healing.
Our findings support the proposal that osteoclasts are the source of OPN in bone cement lines during remodeling.82Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E, Gowen M. Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. → J Bone Miner Res. 1995 Nov;10(11):1666–80. The detection of OPN expression within lacunae represents clear evidence of the secretion of OPN by osteoclasts, because exogenously added OPN has no access to these sites.83Standal T, Borset M, Sundan A. Role of osteopontin in adhesion, migration, cell survival and bone remodeling. → Exp Oncol. 2004 Sep;26(3):179–84.
Conclusion
Immunohistochemical expression of OPN is related to bone remodeling and maturation changes in maxillary sinus lift with ABB xenograft.
Acknowledgments
The authors are grateful to María Dolores Rodríguez Martínez and Jorge A. Payá for technical assistance and to Richard Davies for assistance with the English translation.
Competing interests
The authors declare that they have no competing interests. This investigation was supported in part by Research Groups #CTS-138 and CTS-583 (Regional Government of Andalusia, Spain).
References
| 1. | ↑ | Young MF, Kerr JM, Termine JD, Wewer UM, Wang MG, McBride OW, Fisher LW. cDNA cloning, mRNA distribution and heterogeneity, chromosomal location, and RFLP analysis of human osteopontin (OPN). → Genomics. 1990 Aug;7(4):491–502. |
| 2. | ↑ | Oldberg A, Franzén A, Heinegård D. Cloning and sequence analysis of rat bone sialoprotein (osteopontin) cDNA reveals an Arg-Gly-Asp cell-binding sequence. → Proc Natl Acad Sci USA. 1986 Dec;83(23):8819–23. |
| 3. | ↑ | Hijiya N, Setoguchi M, Matsuura K, Higuchi Y, Akizuki S, Yamamoto S. Cloning and characterization of the human osteopontin gene and its promoter. → Biochem J. 1994 Oct;303(Pt 1):255–62. |
| 4. | ↑ | Milam SB, Haskin C, Zardeneta G, Chen D, Magnuson VL, Klebe RJ. Cell adhesion proteins in oral biology. → Crit Rev Oral Biol Med. 1991;2(4):451–91. |
| 5, 54. | ↑ | Matusan-Ilijas K, Behrem S, Jonjic N, Zarkovic K, Lucin K. Osteopontin expression correlates with angiogenesis and survival in malignant astrocytoma. → Pathol Oncol Res. 2008 Sep;14(3):293–8. |
| 6, 58, 62. | ↑ | Sodek J, Ganss B, McKee MD. Osteopontin. → Crit Rev Oral Biol Med. 2000;11(3):279–303. |
| 7, 18. | ↑ | Taller A, Grohe B, Rogers KA, Goldberg HA, Hunter GK. Specific adsorption of osteopontin and synthetic polypeptides to calcium oxalate monohydrate crystals. → Biophys J. 2007 Sept;93(5):1768–77. |
| 8. | ↑ | Sims NA, Gooi JH. Bone remodeling: multiple cellular interactions required for coupling of bone formation and resorption. → Semin Cell Dev Biol. 2008 Oct;19(5):444–51. |
| 9. | ↑ | Zohar R, Lee W, Arora P, Cheifetz S, McCulloch C, Sodek J. Single cell analysis of intracellular osteopontin in osteogenic cultures of fetal rat calvarial cells. → J Cell Physiol. 1997 Jan;170(1):88–100. |
| 10. | ↑ | McKee MD, Nanci A. Osteopontin and the bone remodeling sequence: colloidal-gold immunocytochemistry of an interfacial extracellular matrix protein. → Ann N Y Acad Sci. 1995 Aug;760:177–89. |
| 11. | ↑ | Sodek J, Chen J, Nagata T, Kasugai S, Todescan R Jr, Li IW, Kim RH. Regulation of osteopontin expression in osteoblasts. → Ann N Y Acad Sci. 1995 Aug;760:223–41. |
| 12. | ↑ | Giachelli CM, Schwartz SM, Liaw L. Molecular and cellular biology of osteopontin: potential role in cardiovascular disease. → Trends Cardiovasc Med. 1995 May-Jun;5(3):88–95. |
| 13, 36, 49, 69, 70, 81. | ↑ | Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. → Clin Oral Implants Res. 2010 Jan;21(1):55–64. |
| 14, 64. | ↑ | Choi ST, Kim JH, Kang EJ, Lee SW, Park MC, Park YB, Lee SK. Osteopontin might be involved in bone remodelling rather than in inflammation in ankylosing spondylitis. → Rheumatology (Oxford). 2008 Dec;47(12):1775–9. |
| 15. | ↑ | Jang JH, Kim JH. Improved cellular response of osteoblast cells using recombinant human osteopontin protein produced by Escherichia coli. → Biotechnol Lett 2005 Nov;27(22):1767–70. |
| 16, 23. | ↑ | Denhardt DT, Noda M. Osteopontin expression and function: role in bone remodeling. → J Cell Biochem Suppl. 1998;30–31 Suppl:92–102. |
| 17. | ↑ | Ishijima M, Ezura Y, Tsuji K, Rittling SR, Kurosawa H, Denhardt DT, Emi M, Nifujia A, Noda M. Osteopontin is associated with nuclear factor κB gene expression during tail-suspension-induced bone loss. → Exp Cell Res. 2006 Oct;312(16):3075–83. |
| 19. | ↑ | Goldberg HA, Warner KJ, Li MC, Hunter GK. Binding of bone sialoprotein, osteopontin and synthetic polypeptides to hydroxyapatite. → Connect Tissue Res. 2001;42(1):25–37. |
| 20. | ↑ | Pampena DA, Robertson KA, Litvinova O, Lajoie G, Goldberg HA, Hunter GK. Inhibition of hydroxyapatite formation by osteopontin phosphopeptides. → Biochem J. 2004 Mar;378(3):1083–7. |
| 21. | ↑ | Altıntaş A, Saruhan-Direskeneli G, Benbir G, Demir M, Purisa S. The role of osteopontin: a shared pathway in the pathogenesis of multiple sclerosis and osteoporosis? → J Neurol Sci. 2009 Jan;276(1–2):41–4. |
| 22. | ↑ | Krause SW, Rehli M, Kreutz M, Schwarzfischer L, Paulauskis JD, Andreesen R. Differential screening identifies genetic markers of monocyte to macrophage maturation. → J Leukoc Biol. 1996 Oct;60(4):540–5. |
| 24. | ↑ | Ihara H, Denhardt DT, Furuya K, Yamashita T, Muguruma Y, Tsuji K, Hruska KA, Higashio K, Enomoto S, Nifuji A, Rittling SR, Noda M. Parathyroid hormoneinduced bone resorption does not occur in the absence of osteopontin. → J Biol Chem. 2001 Apr;276(16):13065–71. |
| 25, 83. | ↑ | Standal T, Borset M, Sundan A. Role of osteopontin in adhesion, migration, cell survival and bone remodeling. → Exp Oncol. 2004 Sep;26(3):179–84. |
| 26, 28. | ↑ | Chellaiah MA, Kizer N, Biswas R, Alvarez U, Strauss-Schoenberger J, Rifas L, Rittling SR, Denhardt DT, Hruska KA. Osteopontin deficiency produces osteoclast dysfunction due to reduced CD44 surface expression. → Mol Biol Cell. 2003 Jan;14(1):173–89. |
| 27. | ↑ | Tani-Ishii N, Tsunoda A, Umemoto T. Osteopontin antisense deoxyoligonucleotides inhibit bone resorption by mouse osteoclasts in vitro. → J Periodontal Res. 1997 Aug;32(6):480–6. |
| 29. | ↑ | Suzuki K, Zhu B, Rittling SR, Denhardt DT, Goldberg HA, McCulloch CA, Sodek J. Colocalization of intracellular osteopontin with CD44 is associated with migration, cell fusion, and resorption in osteoclasts. → J Bone Miner Res. 2002 Aug; 17(8):1486–97. |
| 30. | ↑ | Carinci F, Piattelli A, Degidi M, Palmieri A, Perrotti V, Scapoli L, Martinelli M, Laino G, Pezzetti F. Genetic effects of anorganic bovine bone (Bio-Oss) on osteoblast-like MG63 cells. → Arch Oral Biol. 2006 Feb;51(2):154–63. |
| 31. | ↑ | Tadjoedin ES, De Lange GL, Bronckers AL, Lyaruu DM, Burger EH. Deproteinized cancellous bovine bone (Bio-Oss) as bone substitute for sinus floor elevation: a retrospective, histomorphometrical study of five cases. → J Clin Periodontol. 2003 Mar;30(3):261–70. |
| 32. | ↑ | Yildirim M, Spiekermann H, Biesterfeld S, Edelhoff D. Maxillary sinus augmentation using xenogenic bone substitute material Bio-Oss in combination with venous blood: a histologic and histomorphometric study in humans. → Clin Oral Implants Res. 2000 Jun;11(3):217–29. |
| 33. | ↑ | Schwartz Z, Weesner T, Van Dijk S, Cochran DL, Mellonig JT, Lohmann CH, Carnes DL, Goldstein M, Dean DD, Boyan BD. Ability of deproteinized cancellous bovine bone to induce new bone formation. → J Periodontol. 2000 Aug;71(8):1258–69. |
| 34, 51, 53. | ↑ | Galindo-Moreno P, Padial-Molina M, Fernández-Barbero JE, Mesa F, Rodríguez- Martínez D, O’Valle F. Optimal microvessel density from composite graft of autogenous maxillary cortical bone and anorganic bovine bone in sinus augmentation: influence of clinical variables. → Clin Oral Implants Res. 2010 Feb;21(2):221–7. |
| 35, 59, 66, 82. | ↑ | Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, Dul E, Gowen M. Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. → J Bone Miner Res. 1995 Nov;10(11):1666–80. |
| 37. | ↑ | McKee MD, Farach-Carson MC, Butler WT, Hauschka PV, Nanci A. Ultrastructural immunolocalization of noncollagenous (osteopontin and osteocalcin) and plasma (albumin and α2HS-glycoprotein) proteins in rat bone. → J Bone Miner Res. 1993 Apr;8(4):485–96. |
| 38. | ↑ | Schücklenk U, Ashcroft R. International research ethics. → Bioethics. 2000 Apr;14(2):158–72. |
| 39. | ↑ | American Society of Anesthesiologists. ASA Physical Status Classification System [Internet]. → Schaumburg, Ill.: American Society of Anesthesiologists; [updated 2014 Oct 15; cited 2015 Jul 1]. Available from: http://www.asahq.org/resources/clinicalinformation/ asa-physical-status-classifica tion-system. |
| 40. | ↑ | O’Leary TJ, Drake RB, Naylor JE. The plaque control record. → J Periodontol. 1972 Jan;43(1):38. |
| 41. | ↑ | Wang HL, Katranji A. ABC sinus augmentation classification. → Int J Periodontics Restorative Dent. 2008 Jul-Aug;28(4):383–9. |
| 42. | ↑ | Zinner ID, Small SA. Sinus-lift graft: using the maxillary sinuses to support implants. → J Am Dent Assoc. 1996 Jan;127(1):51–7. |
| 43. | ↑ | Levin L, Herzberg R, Dolev E, Schwartz- Arad D. Smoking and complications of onlay bone grafts and sinus lift operations. → Int J Oral Maxillofac Implants. 2004 May-Jun;19(3):369–73. |
| 44. | ↑ | Galindo-Moreno P, Fauri M, Ávila-Ortiz G, Fernández-Barbero JE, Cabrera-León A, Sánchez-Fernández E. Influence of alcohol and tobacco habits on peri-implant marginal bone loss: a prospective study. → Clin Oral Implants Res. 2005 Oct;16(5):579–86. |
| 45. | ↑ | Galindo-Moreno P, Ávila G, Fernández- Barbero JE, Aguilar M, Sánchez-Fernández E, Cutando A, Wang HL. Evaluation of sinus floor elevation using a composite bone graft mixture. → Clin Oral Implants Res. 2007 Jun;18(3):376–82. |
| 46. | ↑ | Galindo-Moreno P, Ávila G, Fernández- Barbero JE, Mesa F, O’Valle-Ravassa F, Wang HL. Clinical and histologic comparison of two different composite grafts for sinus augmentation: a pilot clinical trial. → Clin Oral Implants Res. 2008 Aug;19(8):755–9. |
| 47. | ↑ | Hallman M, Thor A. Bone substitutes and growth factors as an alternative/complement to autogenous bone for grafting in implant dentistry. → Periodontol 2000. 2008 Jun;47(1):172–92. |
| 48. | ↑ | Wenz B, Oesch B, Horst M. Analysis of the risk of transmitting bovine spongiform encephalopathy through bone grafts derived from bovine bone. → Biomaterials. 2001 Jun;22(12):1599–606. |
| 50. | ↑ | Habibovic P, Yuan H, van der Valk CM, Meijer G, van Blitterswijk CA, de Groot K. 3D microenvironment as essential element for osteoinduction by biomaterials. → Biomaterials. 2005 Jun;26(17):3565–75. |
| 52. | ↑ | Weibrich G, Trettin R, Gnoth SH, Götz H, Duschner H, Wagner W. Bestimmung der Größe der spezifischen Oberfläche von Knochenersatzmaterialien mittels Gasadsorption [Determining the size of the specific surface of bone substitutes with gas adsorption]. → Mund Kiefer Gesichtschir. 2000 May;4(3):148–52. German. |
| 55. | ↑ | Duvall CL, Taylor WR, Weiss D, Wojtowicz AM, Guldberg RE. Impaired angiogenesis, early callus formation, and late stage remodeling in fracture healing of osteopontin-deficient mice. → J Bone Miner Res. 2007 Feb;22(2):286–97. |
| 56. | ↑ | McKee MD, Nanci A. Osteopontin at mineralized tissue interfaces in bone, teeth, and osseointegrated implants: ultrastructural distribution and implications for mineralized tissue formation, turnover, and repair. Microsc Res Tech. 1996 Feb;33(2):141–64. |
| 57. | ↑ | McKee MD, Nanci A. Osteopontin: an interfacial extracellular matrix protein in mineralized tissues. → Connect Tissue Res. 1996 Jan;35(1–4):197–205. |
| 60. | ↑ | McKee MD, Nanci A. Osteopontin deposition in remodeling bone: an osteoblast mediated event. → J Bone Miner Res. 1996 Jun;11(6):873–5. |
| 61. | ↑ | Ganss B, Kim RH, Sodek J. Bone sialoprotein. → Crit Rev Oral Biol Med. 1999;10(1):79–98. |
| 63, 67. | ↑ | Kunii Y, Niwa S, Hagiwara Y, Maeda M, Seitoh T, Suzuki T. The immunohistochemical expression profile of osteopontin in normal human tissues using two site-specific antibodies reveals a wide distribution of positive cells and extensive expression in the central and peripheral nervous systems. → Med Mol Morphol. 2009 Sep;42(3):155–61. |
| 65. | ↑ | Merry K, Dodds R, Littlewood A, Gowen M. Expression of osteopontin mRNA by osteoclasts and osteoblasts in modelling adult human bone. → J Cell Sci. 1993 Apr;104(4):1013–20. |
| 68. | ↑ | Bonewald L. Osteocytes as multifunctional cells. → J Musculoskelet Neuronal Interact. 2006 Oct-Dec;6(4):331–3. |
| 71. | ↑ | Holt I, Marshall MJ. Integrin subunit β3 plays a crucial role in the movement of osteoclasts from the periosteum to the bone surface. → J Cell Physiol. 1998 Apr;175(1):1–9. |
| 72. | ↑ | Chellaiah MA, Hruska KA. The integrin αvβ3 and CD44 regulate the actions of osteopontin on osteoclast motility. → Calcif Tissue Int. 2003 Mar;72(3):197–205. |
| 73. | ↑ | Nakamura I, Pilkington MF, Lakkakorpi PT, Lipfert L, Sims SM, Dixon SJ, Rodan GA, Duong LT. Role of alpha(v)beta(3) integrin in osteoclast migration and formation of the sealing zone. → J Cell Sci. 1999 Nov;112(22):3985–93. |
| 74. | ↑ | Perrotti V, Nicholls BM, Horton MA, Piattelli A. Human osteoclast formation and activity on a xenogenous bone mineral. → J Biomed Mater Res A. 2009 Jun;90A(1):238–46. |
| 75. | ↑ | Nakamura I, Duong LT, Rodan SB, Rodan GA. Involvement of αvβ3 integrins in osteoclast function. → J Bone Miner Metab. 2007 Nov;25(6):337–44. |
| 76. | ↑ | Reinholt FP, Hultenby K, Oldberg A, Heinegård D. Osteopontin—a possible anchor of osteoclasts to bone. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4473–5. |
| 77. | ↑ | Ory S, Brazier H, Pawlak G, Blangy A. Rho GTPases in osteoclasts: orchestrators of podosome arrangement. Eur J Cell Biol. 2008 Sep;87(8-9):469–77. |
| 78. | ↑ | Saad FA, Salih E, Glimcher MJ. Identification of osteopontin phosphorylation sites involved in bone remodeling and inhibition of pathological calcification. J Cell Biochem. 2008 Feb;103(3):852–6. |
| 79. | ↑ | Ek-Rylander B, Andersson G. Osteoclast migration on phosphorylated osteopontin is regulated by endogenous tartrateresistant acid phosphatase. Exp Cell Res. 2010 Feb;316(3):443–51. |
| 80. | ↑ | Schlegel AK, Donath K. BIO-OSS—a resorbable bone substitute? J Long Term Eff Med Implants. 1998;8(3–4):201–9. References 00_JOSR_A4_Moreno_Layout 1 18.09.15 22:17 Seite 9 |






Leave a Reply
Be the First to Comment!