Identification of Staphylococcus aureus at the internal and external implant surfaces in individuals with periimplant disease: A cross-sectional study
March 11, 2016 / Categories: Digital Dentistry, Implant Dentistry

Canullo, Luigi

Orlato Rossetti, Paulo Henrique

Tallarico, Marco

Peñarrocha Oltra, David
The objective of this study was to investigate the prevalence of Staphylococcus aureus (S. aureus) at internal and external dental implant surfaces in patients with periimplant disease.
Introduction
Dental implantology is a central part of modern dentistry concerned with the replacement of missing teeth in various clinical situations.
In the past 30 years, the materials and methods of implant dentistry have undergone a substantial process of development and evolution. Implant surface, macrodesign and type of implant–abutment connection have been found to be of major relevance to initial healing and long-term stability.1Renvert S, Polyzois I, Claffey N. How do implant surface characteristics influence periimplant disease? → J Clin Periodontol. 2011 Mar;38 Suppl 11:214–22.2Bateli M, Att W, Strub JR. Implant neck configurations for preservation of marginal bone level: a systematic review. → Int J Oral Maxillofac Implants. 2011 Mar-Apr;26(2):290–303.3Lang NP, Jepsen S. Implant surfaces and design (Working Group 4). → Clin Oral Implants Res. 2009 Sep;20 Suppl 4:228–31. Since the number of implants placed has increased in the last ten years, optimal maintenance has become increasingly important.4Lang NP, Berglundh T; Working Group 4 of the Seventh European Workshop on Periodontology. Periimplant diseases: where are we now?—Consensus of the Seventh European Workshop on Periodontology. → J Clin Periodontol. 2011 Mar;38 Suppl 11:178–81.5Pjetursson BE, Thoma D, Jung R, Zwahlen M, Zembic A. A systematic review of the survival and complication rates of implantsupported fixed dental prostheses (FDPs) after a mean observation period of at least 5 years. → Clin Oral Implants Res. 2012 Oct;23 Suppl 6:22–38. While in many cases, it has been reported that dental implants are a safe and predictable treatment method with high survival rates, they are not immune from biological and iatrogenic complications associated with improper treatment planning, surgical and prosthetic execution, or material failure, as well as maintenance problems.6Pjetursson BE, Thoma D, Jung R, Zwahlen M, Zembic A. A systematic review of the survival and complication rates of implantsupported fixed dental prostheses (FDPs) after a mean observation period of at least 5 years. → Clin Oral Implants Res. 2012 Oct;23 Suppl 6:22–38. Also, the biological complications of periimplant mucositis and periimplantitis, which may result in soft- and hard-tissue defects, have been suggested to be relevant for later marginal bone loss.7Chan HL, Lin GH, Suarez F, MacEachern M, Wang HL. Surgical management of periimplantitis: a systematic review and meta-analysis of treatment outcomes. → J Periodontol. 2014 Aug;85(8):1027–41.
Several approaches have been followed in seeking to understand the pathomechanism of periimplantitis. According to a consensus conference of the American Academy of Periodontology, bacterial colonization of the implant surface and the occurrence of bone loss indicate the point of no return in periimplantitis.8American Academy of Periodontology. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. → J Periodontol. 2013 Apr;84(4):436–43. Periimplantitis is characterized by an inflammatory process around an implant that includes both soft-tissue inflammation and progressive loss of periimplant supporting bone. Periimplantitis occurs primarily as a result of overwhelming periodontal insult and subsequent immune response.9American Academy of Periodontology. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. → J Periodontol. 2013 Apr;84(4):436–43. The connection to periodontitis as an infectious disease with comparable symptoms and outcomes suggests that investigating the associated local bacteria is fundamental to establishing the pathomechanism of periimplantitis.
The implant surface may be colonized with different pathogens other than periodontal bacteria.10Koyanagi T, Sakamoto M, Takeuchi Y, Maruyama N, Ohkuma M, Izumi Y. Comprehensive microbiological findings in peri-implantitis and periodontitis. → J Clin Periodontol. 2013 Mar;40(3):218–26. According to Albertini et al., opportunistic pathogens such as Pseudomonas aeruginosa, Staphylococcus aureus (S. aureus) and Candida albicans may be associated with implant failure.11Albertini M, López-Cerero L, O’Sullivan MG, Chereguini CF, Ballesta S, Ríos V, Herrero- Climent M, Bullón P. Assessment of periodontal and opportunistic flora in patients with peri-implantitis. → Clin Oral Implants Res. 2015 Aug;26(8):937–41.
As suggested in an American Academy of Periodontology report, secondary diagnostic measures, that is, bacterial culturing, inflammatory markers and genetic factors, may be useful in the diagnosis of periimplant disease.12American Academy of Periodontology. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. → J Periodontol. 2013 Apr;84(4):436–43. According to Canullo et al., bacterial agglomerates around dental implants and their prosthodontic adjacent structures have been identified.13Canullo L, Penarrocha-Oltra D, Soldini C, Mazzocco F, Penarrocha M, Covani U. Microbiological assessment of the implant-abutment interface in different connections: cross-sectional study after 5 years of functional loading. → Clin Oral Implants Res. 2015 Apr;26(4):426–34. These results suggested that all of the connections were contaminated after five years of functional loading; thus, the implant–abutment connection design might influence bacterial activity levels qualitatively and quantitatively, especially inside the implant connection.14Canullo L, Penarrocha-Oltra D, Soldini C, Mazzocco F, Penarrocha M, Covani U. Microbiological assessment of the implant-abutment interface in different connections: cross-sectional study after 5 years of functional loading. → Clin Oral Implants Res. 2015 Apr;26(4):426–34. Furthermore, Cosyn et al. found that intracoronal compartments of screw-retained fixed restorations were heavily contaminated.15Cosyn J, Van Aelst L, Collaert B, Persson GR, De Bruyn H. The periimplant sulcus compared with internal implant and suprastructure components: a microbiological analysis. → Clin Implant Dent Relat Res. 2011 Dec;13(4):286–95. Further investigations have shown that the restorative margin is the principal pathway for bacterial leakage and contamination of abutment screws, and bacteria most likely pass from the periimplant sulcus through the implant–abutment and abutment–prosthesis interfaces.16Canullo L, Penarrocha-Oltra D, Soldini C, Mazzocco F, Penarrocha M, Covani U. Microbiological assessment of the implant-abutment interface in different connections: cross-sectional study after 5 years of functional loading. → Clin Oral Implants Res. 2015 Apr;26(4):426–34.
With the aim of identifying the pathogens that contribute toward the development of periimplantitis defects, different working groups have reported a cluster of bacteria, including Treponema forsythia and S. aureus, associated with periimplant disease.17Persson GR, Renvert S. Cluster of bacteria associated with peri-implantitis. → Clin Implant Dent Relat Res. 2014 Dec;16(6):783–93.
The presence of S. aureus as an opportunistic pathogen in the early stage of active periimplantitis in patients has also been confirmed by Mombelli and Décaillet.18Mombelli A, Décaillet F. The characteristics of biofilms in peri-implant disease. → J Clin Periodontol. 2011 Mar;38 Suppl 11:203–13. In addition, Salvi et al. reported that detection or lack of S. aureus at implant sites at 12 weeks resulted in the highest positive (i.e., 80%) and negative (i.e., 90%) predictive values for the incidence of periimplantitis, respectively.19Salvi GE, Fürst MM, Lang NP, Persson GR. One-year bacterial colonization patterns of Staphylococcus aureus and other bacteria at implants and adjacent teeth. → Clin Oral Implants Res. 2008 Mar;19(3):242–8. Moreover, Canullo et al. showed that S. aureus is present on the external and internal abutment surfaces if these are not cleaned before screwing.20Canullo L, Penarrocha D, Micarelli C, Massidda O, Bazzoli M. Hard tissue response to argon plasma cleaning/sterilisation of customised titanium abutments versus 5-second steam cleaning: results of a 2-year post-loading follow-up from an explanatory randomised controlled trial in periodontally healthy patients. → Eur J Oral Implantol. 2013 Autumn;6(3):251–60.
The aim of the present study is to investigate the prevalence of S. aureus in the oral cavity of patients with active periimplantitis. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.21Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. → J Clin Epidemiol. 2008 Apr;61(4):344–9.
Materials & methods
Study Design
This cross-sectional study evaluated data collected from 51 consecutive, partially edentulous patients of both sexes, aged 18 or older (mean age of 54.2), who had been treated with a single implant-supported, cemented or screw-retained restoration functionally loaded for at least 12 months, with adjacent healthy teeth, but presenting signs of periimplant disease according to Mombelli and Décaillet.22Mombelli A, Décaillet F. The characteristics of biofilms in peri-implant disease. → J Clin Periodontol. 2011 Mar;38 Suppl 11:203–13. The patients were invited to participate and were enrolled after being given a detailed explanation of the study protocol. Written informed consent was obtained for each patient. All of the patients were recruited from the Department of Oral Surgery, University of Valencia, Spain, between September and December 2013. The investigation was conducted according to the principles outlined in the Declaration of Helsinki of 1975 for biomedical research involving human subjects, as amended in 2008. All patients were evaluated clinically and radiographically, and their medical histories were recorded. Bone volumes were analyzed using periapical radiographs.The inclusion criteria were:
- presence of periimplant disease with a vertical bone defect of > 3 mm after implant integration according to Mombelli and Décaillet.23Mombelli A, Décaillet F. The characteristics of biofilms in peri-implant disease. → J Clin Periodontol. 2011 Mar;38 Suppl 11:203–13.
- age > 18.
- no relevant medical conditions.
The exclusion criteria were:
- pregnancy or lactation.
- known systemic disease or metabolic disorders (e.g., HIV) treated with medication detrimental to soft tissue and/or bone healing (e.g., high-dose steroid therapy, systemic treatment with tetracycline or tetracycline analogs, bone therapeutic levels of fluorides, bisphosphonates, medication affecting bone turnover, antibiotics for more than seven days or any investigational drug)—topical application of steroids and steroid application through inhalation were not exclusion criteria.
- malignant diseases or other diseases treated with radiotherapy or chemotherapeutic agents (chemotherapy) during the past five years.
- a history of head and neck radiation treatment owing to certain medical conditions.
- a suspected allergy or incompatibility with any of the bone graft substitute components (calcium phosphates, PLGA, NMP).
- inability to comply with the protocol requirements, including severe alcohol or drug user.
- involvement in any other clinical trial during the course of the present trial, or within a period of 30 days prior to its beginning or after its completion.
- acute abscesses localized in the proximity of the prospective surgical field.
After full screening, 16 patients were to be excluded: 13 had taken systemic antibiotics during the three months prior to the microbiological sampling, two were pregnant, and one refused to participate. The final sample consisted of 35 individuals (20 male, 15 female) and 63 affected dental implants.
Microbiological sampling
Samples for microbiological analysis were obtained from four sites in each patient in the following order: (1) the periimplant sulcular fluid (PISF) of each implant; (2) the gingival sulcus (GS) of the adjacent teeth, used as control group; (3) the inner portions of the implant connection and the abutment of each implant (IIP); and (4) the oropharyngeal complex (OF). In all of the groups, the microbiological samples were taken using the GUIDOR Perio-Implant Diagnostic Kit (Sunstar Iberia, Sant Just Desvern, Spain), consisting of five sterile absorbent paper tips and an empty sterile 2 ml microtube.
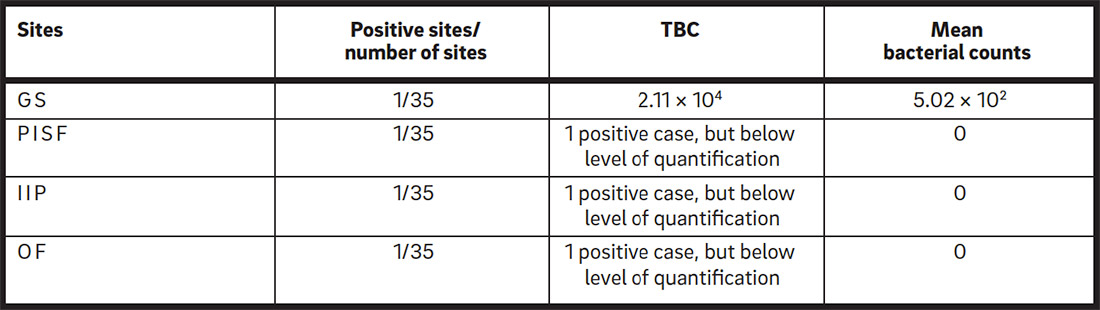
Investigated sites and bacterial counts.
GS: gingival sulcus of the adjacent teeth; PISF: periimplant sulcular fluid; IIP: inner parts of connection; OF: oropharyngeal complex.
Prior to collection of the subgingival plaque, supragingival plaque was eliminated from implants and teeth using a cotton tip, without penetrating the GS. OptraGate (Ivoclar Vivadent, Schaan, Liechtenstein) was used to retract the lips and cheeks completely and to ensure relative isolation. After light drying of the area with an air syringe, paper tips were inserted into the periimplant or periodontal sulci for 30 s. The samples from the inner surfaces of the implant–abutment complex were obtained after careful removal of both the restorations and the abutments, seeking to avoid contamination. One drop of RNA- and DNA-free water (Water Molecular Biology Reagent, W4502, Sigma-Aldrich, St. Louis, Mo., U.S.) was placed inside the implant connection and three paper tips were inserted for 30 s. The inner surface of the abutment was wet with a drop of RNA- and DNA-free water and smeared with two paper tips. The paper tips were placed into the microtubes and sent for microbiological analysis to the Institut Clinident laboratory (Aix-en-Provence, France) in the provided mailing envelopes. Finally, an oral environment analysis was performed using sterile cotton swabs collected from the cheeks, tongue, throat and pharynx of each patient.
After sample collection, the inner part of the implants and the abutment–restoration complex were cleaned with a 5% chlorhexidine solution in an ultrasonic bath for 10 min. Afterward, a new abutment screw was connected using a torque wrench (Torq Control, Anthogyr, Sallanches, France) until it reached a torque of 30 N cm, according to the manufacturer’s instructions. In order to verify proper fit between the dental restoration and the implant, standardized digital periapical radiographs were taken using a dental radiographic film holder (Rinn Centrator Bite, DENTSPLY RINN, Elgin, Ill., U.S.) and the paralleling technique.
Quantitative real-time polymerase chain reaction assays
Quantitative real-time polymerase chain reaction (PCR) assays were carried out for total bacterial counts (TBCs) for each target species,24Canullo L, Rossetti P, Rakic M, Penarrocha D, Monreal A, Covani U. Treatment of technical and biological complications: opportunistic pathogens (E. faecalis and P. aeruginosa) at internal and external implant portions in peri-implant disease: a cross-sectional study. → Clin Oral Implants Res. 2014 Sep;25 Suppl 10:55.25Penarrocha-Oltra D, Rossetti PH, Covani U, Galluccio F, Canullo L. Microbial leakage at the implant/abutment connection due to implant insertion maneuvers: cross-sectional study 5 years post loading in healthy patients. → J Oral Implantol. 2014 Dec 23. [Epub ahead of print]. in a volume of 10 μL composed of 1× QuantiFast SYBR Green PCR (Qiagen, Hilden, Germany), 2 μL of DNA extract, and 1 μM of each primer. The species-specific PCR primers used in this study were provided by Institut Clinident and manufactured by metabion (Martinsried, Germany). Assays were carried out on the Rotor-Gene Q thermal cycling system (Qiagen) with the following program: for TBC, 95 °C for 30 s, followed by 40 cycles of 10 s at 95 °C, 10 s at 60 °C, and 35 s at 72 °C; for S.aureus, 95 °C for 5 min, followed by 40 cycles of 10 s at 95 °C, 10 s at 66 °C, and 35 s at 72 °C. A final melting curve analysis(70–95 °C in 1 °C steps for 5 s increments) was performed. Fluorescence signals were measured every cycle at the end of the extension step and continuously during the melting curve analysis. Serial dilutions of standard DNA, provided by Institut Clinident, were used in each reaction as external standards for absolute quantification of the target pathogen. Finally, the data were analyzed using Rotor-Gene Q Series Software (Qiagen).
Statistical analysis
The mean and standard deviations for TBCs at each inspected site (PISF, GS, IIP, OF) were recorded and analyzed according to a pre-established analysis plan. A bio-statistician with expertise in dentistry analyzed the data using statistical software (SigmaPlot, Version 13, Systat Software, San Jose, Calif., U.S.). Before running the statistical analysis, the TBCs for each site were transformed (log transformation [log10]) in an attempt to render less skewed distributions, making the data more interpretable and helping to meet the assumptions of inferential statistics. As the normality test failed, a nonparametric test (Kruskal–Wallis) was used. The level of significance was set at α = 0.05.
Results
No implants were lost, and all of the prostheses were in situ at the time of examination. At the end of the study, just one site (out of 35) in the GS of the adjacent teeth presented a TBC of 2.11 × 104. The mean bacterial count of S. aureus was 5.02 × 102; therefore, this value was taken as control. Conversely, in the PISF of each implant, the IIP and the OF complex, the mean bacterial counts of S. aureus were 0, with only one site (out of 35) positive, but below the level of quantification. The data are reported in (Table1). No statistically significant differences were found among groups regarding site location (Kruskal–Wallis test; p = 0.40).
Discussion
Currently, there are neither standardized antibiotic prophylactic regimens for dental implant placement nor universally accepted treatment for periimplantitis. The treatment of infected implants is difficult and usually requires removal.26Penarrocha-Oltra D, Rossetti PH, Covani U, Galluccio F, Canullo L. Microbial leakage at the implant/abutment connection due to implant insertion maneuvers: cross-sectional study 5 years post loading in healthy patients. → J Oral Implantol. 2014 Dec 23. [Epub ahead of print]. However, it has become clear that therapy of periimplant mucositis should be considered a preventive measure for the onset of periimplantitis. Completion of active periodontal therapy should precede implant placement in periodontally compromised patients.27Canullo L, Rossetti PH, Penarrocha D. Identification of Enterococcus Faecalis and Pseudomonas Aeruginosa on and in implants in individuals with peri-implant disease: a cross-sectional study. → Int J Oral Maxillofac Implants. 2015 May-Jun;30(3):583–7.
S. aureus is a facultative coccus and Gram-positive bacterium normally associated with surgical wounds in orthopedic patients.28Salvi G, Ramseier CA. Efficacy of patient-administered mechanical and/or chemical plaque control protocols in the management of peri-implant mucositis. A systematic review. → J Clin Periodontol. 2015 Apr;42 Suppl 16:S187–201. Part of this can be explained by the impedance seen on cultured osteoblasts, with S. aureus surviving up to 48 h after internalization by those bone cells and still eliciting interleukin 6 and interleukin 8 responses,29Arciola CR, Cervellati M, Pirini V, Gamberini S, Montanaro L. Staphylococci in orthopaedic surgical wounds. → New Microbiol. 2001 Oct;24(4):365–9. which have pro-inflammatory effects and are involved in osteoclastogenesis30Jauregui CE, Mansell JP, Jepson MA, Jenkinson HF. Differential interactions of Streptococcus gordonii and Staphylococcus aureus with cultured osteoblasts. → Mol Oral Microbiol. 2013 Aug;28(4):250–66. and foreign body reactions.31Tuan RS, Lee FYI, Konttinen YT, Wilkinson JM, Smith RL. What are the local and systemic biologic reactions and mediators to wear debris, and what host factors determine or modulate the biologic response to wear particles? → J Am Acad Orthop Surg. 2008 Jul;16 Suppl 1:S42–8. In addition, S. aureus has the ability to form a biofilm and lead to chronic infection.32Trindade R, Albrektsson T, Tengvall P, Wennerberg A. Foreign body reaction to biomaterials: on mechanisms for build-up and breakdown of osseointegration. → Clin Implant Dent Relat Res. 2014 Sep 25. doi:10.1111/cid.12274. [Epub ahead of print].
A retrospective study has demonstrated that patients capable of maintaining high immuno-globulin G antibody titers to S. aureus had successful implants compared with nonosseointegrated fixtures.33Rams TE, Feik D, Slots J. Staphylococci in human periodontal diseases. → Oral Microbiol Immunol. 1990 Feb;5(1):29–32.
In the present study, the lack of significance regarding the bacterial counts of S. aureus at IIP and PISF must be considered, since in vitro this pathogen has shown an affinity for titanium sur- faces,34Prabhakara R, Harro JM, Leid JG, Keegan AD, Prior ML, Shirtliff ME. Suppression of the inflammatory immune response prevents the development of chronic infection due to methicillin resistant Staphylococcus aureus. → Infect Immun. 2011 Dec;79(12):5010–8. and two studies have related its levels to deep periimplant pockets with bleeding on probing.35Harris LG, Mead L, Müller-Oberländer E, Richards RG. Bacteria and cell cytocompatibility studies on coated medical grade titanium surfaces. → J Biomed Mater Res A. 2006 Jul;78A(1):50–8.36Rams TE, Feik D, Slots J. Staphylococci in human periodontal diseases. → Oral Microbiol Immunol. 1990 Feb;5(1):29–32. One study has demonstrated that the bacterial counts of S. aureus increase from 5% to 15% at implant sites 12 weeks after surgery.37Renvert S, Lindahl C, Renvert H, Persson GR. Clinical and microbiological analysis of subjects treated with Brånemark or AstraTech implants: a 7-year follow-up study. → Clin Oral Implants Res. 2008 Apr;19(4):342–7. However, another study pointed out that even after seven years of follow-up the presence of S. aureus at tooth sites could be indicative of the presence of the same pathogen at implant sites,38Rams TE, Feik D, Slots J. Staphylococci in human periodontal diseases. → Oral Microbiol Immunol. 1990 Feb;5(1):29–32. while another study indicated that the lack of S. aureus at implant sites after 12 weeks demonstrated a high negative predictive value after 12 months.39Fürst MM, Salvi GE, Lang NP, Persson GR. Bacterial colonization immediately after installation on oral titanium implants. → Clin Oral Implants Res. 2007 Aug;18(4):501–8. More recently, an article demonstrated that regardless of health status, periodontal and periimplant sites harbored S. aureus cells, being the highest load of all six species analyzed.40Zhuang LF, Watt RM, Mattheos N, Si MS, Lai HC, Lang NP. Periodontal and peri- implant microbiota in patients with healthy and inflamed periodontal and peri-implant tissues. → Clin Oral Implants Res. 2014 Nov 14. doi:10.1111/clr.12508. [Epub ahead of print].
Conclusion
Within the limits of this study, S. aureus could not be quantified inside and around dental implants in detectable limits. However, clinicians must bear in mind that, in the early stage of healing, this pathogen can influence the immune response and lead to periimplant bone loss.
Competing interests
The study was supported by Sweden & Martina (Due Carrare, Italy), which paid for the kits, and by the Institut Clinident, which performed the analysis free of charge. The authors declare that they have no competing interests related to this study.
Acknowledgments
The authors highly appreciate the skills and commitment of Dr. Audrenn Gautier in the supervision of the study. Additionally, the authors wish to offer their gratitude to the Institut Clinident for their professional support in the microbiological analysis.
References
| 1. | ↑ | Renvert S, Polyzois I, Claffey N. How do implant surface characteristics influence periimplant disease? → J Clin Periodontol. 2011 Mar;38 Suppl 11:214–22. |
| 2. | ↑ | Bateli M, Att W, Strub JR. Implant neck configurations for preservation of marginal bone level: a systematic review. → Int J Oral Maxillofac Implants. 2011 Mar-Apr;26(2):290–303. |
| 3. | ↑ | Lang NP, Jepsen S. Implant surfaces and design (Working Group 4). → Clin Oral Implants Res. 2009 Sep;20 Suppl 4:228–31. |
| 4. | ↑ | Lang NP, Berglundh T; Working Group 4 of the Seventh European Workshop on Periodontology. Periimplant diseases: where are we now?—Consensus of the Seventh European Workshop on Periodontology. → J Clin Periodontol. 2011 Mar;38 Suppl 11:178–81. |
| 5, 6. | ↑ | Pjetursson BE, Thoma D, Jung R, Zwahlen M, Zembic A. A systematic review of the survival and complication rates of implantsupported fixed dental prostheses (FDPs) after a mean observation period of at least 5 years. → Clin Oral Implants Res. 2012 Oct;23 Suppl 6:22–38. |
| 7. | ↑ | Chan HL, Lin GH, Suarez F, MacEachern M, Wang HL. Surgical management of periimplantitis: a systematic review and meta-analysis of treatment outcomes. → J Periodontol. 2014 Aug;85(8):1027–41. |
| 8, 9, 12. | ↑ | American Academy of Periodontology. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. → J Periodontol. 2013 Apr;84(4):436–43. |
| 10. | ↑ | Koyanagi T, Sakamoto M, Takeuchi Y, Maruyama N, Ohkuma M, Izumi Y. Comprehensive microbiological findings in peri-implantitis and periodontitis. → J Clin Periodontol. 2013 Mar;40(3):218–26. |
| 11. | ↑ | Albertini M, López-Cerero L, O’Sullivan MG, Chereguini CF, Ballesta S, Ríos V, Herrero- Climent M, Bullón P. Assessment of periodontal and opportunistic flora in patients with peri-implantitis. → Clin Oral Implants Res. 2015 Aug;26(8):937–41. |
| 13, 14, 16. | ↑ | Canullo L, Penarrocha-Oltra D, Soldini C, Mazzocco F, Penarrocha M, Covani U. Microbiological assessment of the implant-abutment interface in different connections: cross-sectional study after 5 years of functional loading. → Clin Oral Implants Res. 2015 Apr;26(4):426–34. |
| 15. | ↑ | Cosyn J, Van Aelst L, Collaert B, Persson GR, De Bruyn H. The periimplant sulcus compared with internal implant and suprastructure components: a microbiological analysis. → Clin Implant Dent Relat Res. 2011 Dec;13(4):286–95. |
| 17. | ↑ | Persson GR, Renvert S. Cluster of bacteria associated with peri-implantitis. → Clin Implant Dent Relat Res. 2014 Dec;16(6):783–93. |
| 18, 22, 23. | ↑ | Mombelli A, Décaillet F. The characteristics of biofilms in peri-implant disease. → J Clin Periodontol. 2011 Mar;38 Suppl 11:203–13. |
| 19. | ↑ | Salvi GE, Fürst MM, Lang NP, Persson GR. One-year bacterial colonization patterns of Staphylococcus aureus and other bacteria at implants and adjacent teeth. → Clin Oral Implants Res. 2008 Mar;19(3):242–8. |
| 20. | ↑ | Canullo L, Penarrocha D, Micarelli C, Massidda O, Bazzoli M. Hard tissue response to argon plasma cleaning/sterilisation of customised titanium abutments versus 5-second steam cleaning: results of a 2-year post-loading follow-up from an explanatory randomised controlled trial in periodontally healthy patients. → Eur J Oral Implantol. 2013 Autumn;6(3):251–60. |
| 21. | ↑ | Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. → J Clin Epidemiol. 2008 Apr;61(4):344–9. |
| 24. | ↑ | Canullo L, Rossetti P, Rakic M, Penarrocha D, Monreal A, Covani U. Treatment of technical and biological complications: opportunistic pathogens (E. faecalis and P. aeruginosa) at internal and external implant portions in peri-implant disease: a cross-sectional study. → Clin Oral Implants Res. 2014 Sep;25 Suppl 10:55. |
| 25, 26. | ↑ | Penarrocha-Oltra D, Rossetti PH, Covani U, Galluccio F, Canullo L. Microbial leakage at the implant/abutment connection due to implant insertion maneuvers: cross-sectional study 5 years post loading in healthy patients. → J Oral Implantol. 2014 Dec 23. [Epub ahead of print]. |
| 27. | ↑ | Canullo L, Rossetti PH, Penarrocha D. Identification of Enterococcus Faecalis and Pseudomonas Aeruginosa on and in implants in individuals with peri-implant disease: a cross-sectional study. → Int J Oral Maxillofac Implants. 2015 May-Jun;30(3):583–7. |
| 28. | ↑ | Salvi G, Ramseier CA. Efficacy of patient-administered mechanical and/or chemical plaque control protocols in the management of peri-implant mucositis. A systematic review. → J Clin Periodontol. 2015 Apr;42 Suppl 16:S187–201. |
| 29. | ↑ | Arciola CR, Cervellati M, Pirini V, Gamberini S, Montanaro L. Staphylococci in orthopaedic surgical wounds. → New Microbiol. 2001 Oct;24(4):365–9. |
| 30. | ↑ | Jauregui CE, Mansell JP, Jepson MA, Jenkinson HF. Differential interactions of Streptococcus gordonii and Staphylococcus aureus with cultured osteoblasts. → Mol Oral Microbiol. 2013 Aug;28(4):250–66. |
| 31. | ↑ | Tuan RS, Lee FYI, Konttinen YT, Wilkinson JM, Smith RL. What are the local and systemic biologic reactions and mediators to wear debris, and what host factors determine or modulate the biologic response to wear particles? → J Am Acad Orthop Surg. 2008 Jul;16 Suppl 1:S42–8. |
| 32. | ↑ | Trindade R, Albrektsson T, Tengvall P, Wennerberg A. Foreign body reaction to biomaterials: on mechanisms for build-up and breakdown of osseointegration. → Clin Implant Dent Relat Res. 2014 Sep 25. doi:10.1111/cid.12274. [Epub ahead of print]. |
| 33, 36, 38. | ↑ | Rams TE, Feik D, Slots J. Staphylococci in human periodontal diseases. → Oral Microbiol Immunol. 1990 Feb;5(1):29–32. |
| 34. | ↑ | Prabhakara R, Harro JM, Leid JG, Keegan AD, Prior ML, Shirtliff ME. Suppression of the inflammatory immune response prevents the development of chronic infection due to methicillin resistant Staphylococcus aureus. → Infect Immun. 2011 Dec;79(12):5010–8. |
| 35. | ↑ | Harris LG, Mead L, Müller-Oberländer E, Richards RG. Bacteria and cell cytocompatibility studies on coated medical grade titanium surfaces. → J Biomed Mater Res A. 2006 Jul;78A(1):50–8. |
| 37. | ↑ | Renvert S, Lindahl C, Renvert H, Persson GR. Clinical and microbiological analysis of subjects treated with Brånemark or AstraTech implants: a 7-year follow-up study. → Clin Oral Implants Res. 2008 Apr;19(4):342–7. |
| 39. | ↑ | Fürst MM, Salvi GE, Lang NP, Persson GR. Bacterial colonization immediately after installation on oral titanium implants. → Clin Oral Implants Res. 2007 Aug;18(4):501–8. |
| 40. | ↑ | Zhuang LF, Watt RM, Mattheos N, Si MS, Lai HC, Lang NP. Periodontal and peri- implant microbiota in patients with healthy and inflamed periodontal and peri-implant tissues. → Clin Oral Implants Res. 2014 Nov 14. doi:10.1111/clr.12508. [Epub ahead of print]. |






Leave a Reply
Be the First to Comment!