Individualized CAD/CAM-produced titanium scaffolds for alveolar bone augmentation: A retrospective analysis of dehiscence events in relation to demographic and surgical parameters
May 8, 2018 / Categories: Digital Dentistry, Implant Dentistry

Seiler, Marcus

Peetz, Michael

Hartmann, Amely

Witkowski, Radoslaw
Abstract
Objectives
Computer-aided design/computer-aided manufacturing (CAD/CAM) technologies may improve application of titanium scaffolds, onlay techniques and guided bone regeneration. In this study, the clinical outcome of DICOM-based individualized CAD/CAM-produced titanium scaffolds (iCTSs) was analyzed in grafted defects, particularly with regard to relation of dehiscence to demographic and surgery-related factors.
Materials and methods
In 100 patients, 115 defects of the alveolar crest were reconstructed with an iCTS covered with a native bilayer collagen membrane or left uncovered. The volume was mostly grafted with a mixture of autogenous bone and deproteinized bovine bone mineral. The healing process was documented. Office records were analyzed for association of dehiscence with demographic and surgical parameters.
Results
Uneventful healing was observed in 82 defects. Infection of the surgical area was documented in 11 cases, 10 were resolved by medication. One defect had to be regrafted. Dehiscence was reported in 26 defects. Premature removal of exposed iCTSs was not necessary. All of the cases showed sufficiently grafted volume for implant placement with presurgical 3-D planning. The grafted volume in the defects with dehiscence did not differ from that in sites without dehiscence. Statistical analysis revealed no significant association of dehiscence with demographic or surgical parameters, but a tendency to higher prevalence of dehiscence with mesiodistal width of the defect.
Conclusion
Combination of an iCTS with guided bone regeneration offers a reliable grafting technique with low sensitivity to dehiscence. Dehiscence did not correlate with demographic or surgical factors. In addition, it did not affect the final outcome, as implant insertion was possible simultaneously or staged in all of the cases.
Keywords
CAD/CAM, individual titanium scaffold, augmentation, wound dehiscence.
Introduction
Replacement of lost teeth with implants is a routine and effective treatment showing high survival rates after long-term monitoring.1Barker D. Implant assessment. → Dent Update. 2012 Mar;39(2):128–32, 134. 2Moraschini V, Poubel LA, Ferreira VF, Barboza Edos S. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: a systematic review. → Int J Oral Maxillofac Surg. 2015 Mar;44(3):377–88. In order to achieve adequate functional and esthetic outcomes, an optimal 3-D implant position has to be assured.3Buser D, Martin W, Belser UC. Optimizing esthetics for implant restorations in the anterior maxilla: anatomic and surgical considerations. → Int J Oral Maxillofac Implants. 2004;19 Suppl:43–61. In many cases, the residual bone width, height and ridge contour are not sufficient for optimal implant placement.4Rasia-dal Polo M, Poli PP, Rancitelli D, Beretta M, Maiorana C. Alveolar ridge reconstruction with titanium meshes: a systematic review of the literature. → Med Oral Patol Oral Cir Bucal. 2014 Nov;19(6):e639–46. 5Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. Therefore, ridge augmentation is recommended in order to maintain the alveolar ridge and simplify subsequent treatment procedures.6Hämmerle CH, Araújo MG, Simion M; Osteology Consensus Group 2011. Evidence-based knowledge on the biology and treatment of extraction sockets. → Clin Oral Implants Res. 2012 Feb;23 Suppl 5:80–2. Despite the availability of various augmentation procedures and materials, the restoration of an adequate amount of bone remains challenging.
The use of titanium scaffolds in terms of guided bone regeneration is a widespread procedure for horizontal and vertical ridge augmentation.7Rasia-dal Polo M, Poli PP, Rancitelli D, Beretta M, Maiorana C. Alveolar ridge reconstruction with titanium meshes: a systematic review of the literature. → Med Oral Patol Oral Cir Bucal. 2014 Nov;19(6):e639–46. Clinical and histological analysis has revealed increased morphological ridge repair and bone density after application of titanium scaffolds together with deproteinized bovine bone mineral (DBBM).8Maiorana C, Santoro F, Rabagliati M, Salina S. Evaluation of the use of iliac cancellous bone and anorganic bovine bone in the reconstruction of the atrophic maxilla with titanium mesh: a clinical and histologic investigation. → Int J Oral Maxillofac Implants. 2001 May–Jun;16(3):427–32.9Artzi Z, Dayan D, Alpern Y, Nemcovsky CE. Vertical ridge augmentation using xenogenic material supported by a configured titanium mesh: clinicohistopathologic and histochemical study. → Int J Oral Maxillofac Implants. 2003 May–Jun;18(3):440–6. Larger vertical gain in ridge can be achieved using titanium scaffolds.10Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. The main disadvantage of prefabricated titanium scaffolds is the intraoperative and time-consuming manual 3-D trimming according to the individual defect size of the patient.11Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8.12Ciocca L, Ragazzini S, Fantini M, Corinaldesi G, Scotti R. Work flow for the prosthetic rehabilitation of atrophic patients with a minimal-intervention CAD/CAM approach. → J Prosthet Dent. 2015 Jul;114(1):22–6. Computer-aided design/computer-aided manufacturing (CAD/CAM) technology can be used to overcome these disadvantages. Using individual patient computed tomography (CT) or cone beam computed tomography (CBCT), the necessary augmentation volume for the defects can be estimated preoperatively. According to the calculated augmentation volume, DICOM-based individualized CAD/CAM-produced titanium scaffolds (iCTSs) can be used to avoid intraoperative trimming of the prefabricated titanium scaffolds. Application of an iCTS shortens surgery time and may reduce the overall costs of the implantation procedure.13Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. 14Ciocca L, Ragazzini S, Fantini M, Corinaldesi G, Scotti R. Work flow for the prosthetic rehabilitation of atrophic patients with a minimal-intervention CAD/CAM approach. → J Prosthet Dent. 2015 Jul;114(1):22–6. The most common complication of titanium scaffolds is flap dehiscence.15Hämmerle CH, Araújo MG, Simion M; Osteology Consensus Group 2011. Evidence-based knowledge on the biology and treatment of extraction sockets. → Clin Oral Implants Res. 2012 Feb;23 Suppl 5:80–2. 16Maiorana C, Santoro F, Rabagliati M, Salina S. Evaluation of the use of iliac cancellous bone and anorganic bovine bone in the reconstruction of the atrophic maxilla with titanium mesh: a clinical and histologic investigation. → Int J Oral Maxillofac Implants. 2001 May–Jun;16(3):427–32. Recently, Sagheb et al. reported on the use of the iCTS for alveolar ridge augmentation and found no negative impact of dehiscence on the outcome.17Sagheb K, Schiegnitz E, Moergel M, Walter C, Al-Nawas B, Wagner W. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. → Int J Implant Dent. 2017 Dec;3(1):36. doi: 10.1186/ s40729-017-0097-z. The objective of this study was to retrospectively analyze the dehiscence rate after iCTS augmentation procedures regarding various demographic and surgery-related factors in 100 patients with 115 bony defects.
Materials and methods
Study design and patient population
The retrospective analysis included patients who underwent implant therapy with additional augmentation procedures between 2014 and 2015 in a clinic for oral and maxillofacial surgery in Filderstadt, Germany. Screening of patients who needed bone augmentation before implantation was done during regular implant consultation. Cases with indication of onlay technique together with a titanium scaffold were evaluated. All of the patients were informed about the different augmentation possibilities. One hundred patients (56 male, 44 female) decided on the Yxoss CBR system (ReOss, Filderstadt, Germany), which provides an iCTS based on the CT/CBCT DICOM data of each patient. CT/CBCT was performed for all of the patients within 3 months before surgery. The production of an iCTS took 2–4 weeks. Each iCTS was controlled and finalized via an internet-based platform provided by the supplier. Using CT/CBCT, the necessary augmentation volume for the defect was calculated and documented. In addition, demographic (age, sex, smoking, periodontitis history) and surgical parameters (region, defect size, flap design, gingival morphotype, graft volume, use of membrane) were recorded. The gingival morphotype was classified into thin gingival morphotype A1 or A2 and into thick gingival morphotype B as follows:18Müller HP, Eger T. Gingival phenotypes in young male adults. → J Clin Periodontol. 1997 Jan;24(1):65–71.
- A1: high-scalloped, gingival thickness of < 1 mm, gingival width of < 3.5 mm, oval tooth form.
- A2: high-scalloped, gingival thickness of < 1 mm, gingival width of < 4–5 mm, oval tooth form.
- B: low-scalloped, gingival thickness of > 1 mm, gingival width of > 6 mm, square tooth form.
Surgical procedure
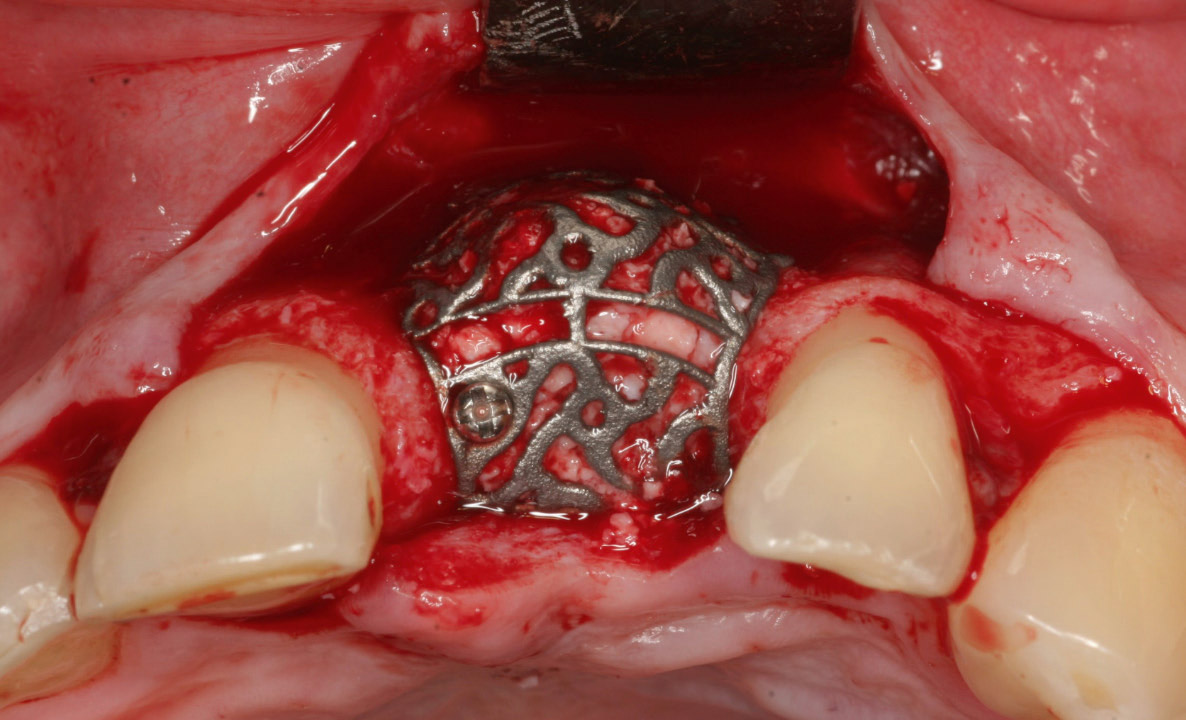
Fig. 1
Intraoperative view of a DICOM-based individualized CAD/CAM-produced titanium scaffold after loading with grafting material and placement on the defect. The iCTS was fixated with a minimum of 1 bone screw.
Surgery was performed under general anesthesia or under local anesthesia. Intraoperative defect assessment was performed using a poncho flap with a deep vestibular incision, a midcrestal ridge incision, a split-thickness flap or the tunnel technique.19Hoexter DL, Epstein SB. The Poncho Flap (repositioned perforated attached gingival flap). → Oral Implantol. 1975 Spring;5(4):547–52. In some cases, an additional palatal rotational flap was performed. For augmentation, a combination of a deproteinized bovine bone mineral (DBBM; Geistlich Bio-Oss granules, 1–2 mm; Geistlich Biomaterials, Wolhusen, Switzerland) and autogenous bone in a ratio of 1:1 (n = 104), autogenous bone alone (n = 2), bone substitute material (KNE) alone (n = 5), an allograft (n = 1) or no material (n = 1) was used. Autogenous bone was harvested from the retromolar region using a hollow trephine drill with an inner diameter of 6 mm, followed by grinding of the small bone cylinders in a bone mill (Bull Bone Mill, MONDEAL Medical Systems, Mühlheim an der Donau, Germany). The iCTS was loaded with grafting material, placed on the defect and fixated with a minimum of 1 bone screw (Fig. 1). The iCTS was covered in situ with a porcine native bilayer collagen membrane (Geistlich Bio-Gide, Geistlich Biomaterials; n = 79) or left uncovered (n = 35). The surgical area was completely closed and the flap fixed with mattress, sling or single sutures. After surgery, antibiotics were prescribed orally for 5 days (amoxicillin, 1,000 mg, 1-0-1). Radiographic control of the graft and the iCTS was performed using CBCT after healing. Postsurgery, patients were recalled for follow-ups to control the surgical area for wound dehiscence and inflammation. Removal of the iCTS and simultaneous implantation were performed depending on the healing period (5–8 months) postsurgery. The same surgical approach was used for the baseline defect assessment. After loosening of the fixing screw, the iCTS was carefully removed by applying rotating forces to predetermined breaking points of the summit of the iCTS at the top of the scaffold with a standard periosteal elevator. The surgical area was closed without tension and the sutures removed 1 week after surgery. Implants were placed simultaneously (n = 63) with augmentation or after 5–8 months (n = 50).
Statistical data analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows (Version 21.0, IBM, Armonk, N.Y., U.S.). Quantitative data (augmentation volume) were descriptively analyzed for arithmetic mean and standard deviation. For comparison of augmented volumes in the defect sites with and without dehiscence, the Mann– Whitney U test was used because the data were not normally distributed (according to the Shapiro–Wilk test, P < 0.001). Association of dehiscence with demographic (age, sex, smoking, periodontitis history) and surgical parameters (gingival morphotype, surgical access, region, use of membrane) were analyzed using the chisquared test or Fisher exact test (if cell occupancy numbers were < 5). Two-sample tests were performed. The impact of predictive factors on the risk of dehiscence was investigated using univariate and multivariate models for logistic regression analysis. Results were considered to be statistically significant if the P value was ≤ 0.05. Adaptation for multiple testing was not performed, since the analysis was explorative and used to test the hypothesis. Nonconsideration of intrapatient correlations influencing P values might have influenced the statistical analysis.
Results
Patient population and description of defect sites
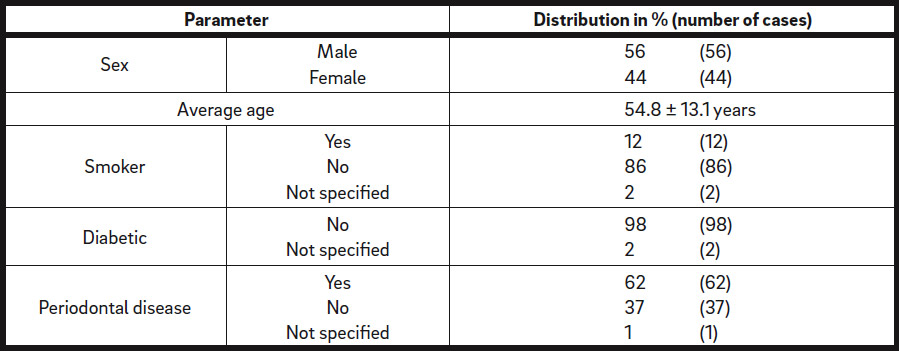
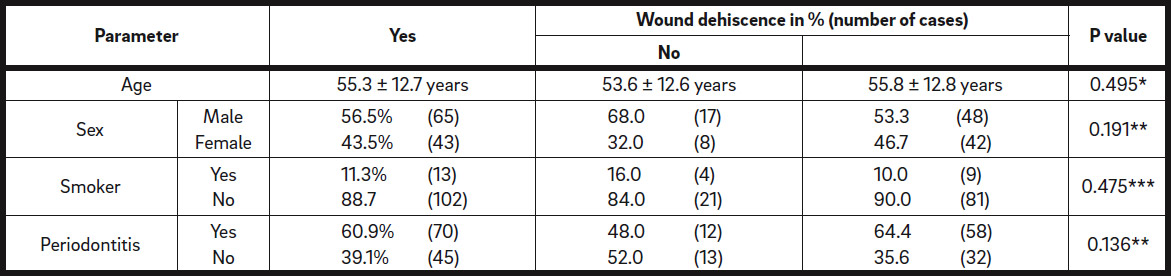
One hundred patients with 115 defect regions in total were retrospectively analyzed. Of these patients, 56 were male and 44 female, with an average age of 54.8 ± 13.1 years (range: 18–82 years; Table 1). Twelve of the patients were smokers (12%). Sixty-two patients presented with periodontitis (62%; Table 1) and were treated by the referring dentist before surgical intervention.
Demographics and patient history. Total number of patients: N = 100, with 115 defect regions.
Description of defect regions and surgical procedure. Total number of defect regions: n = 115.
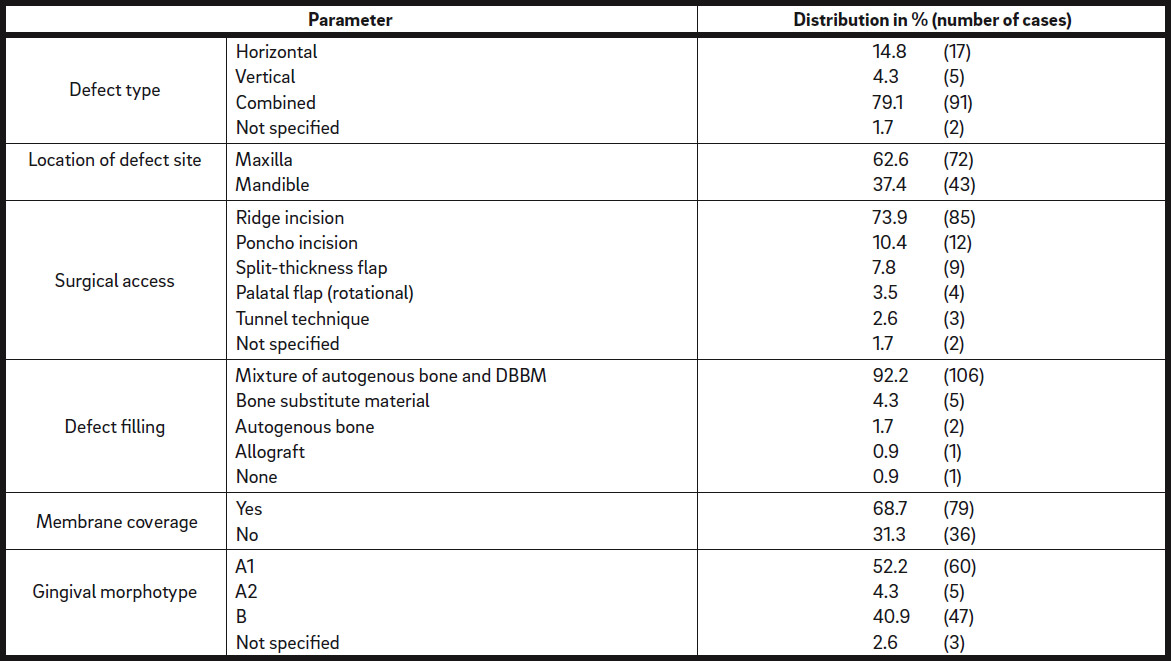
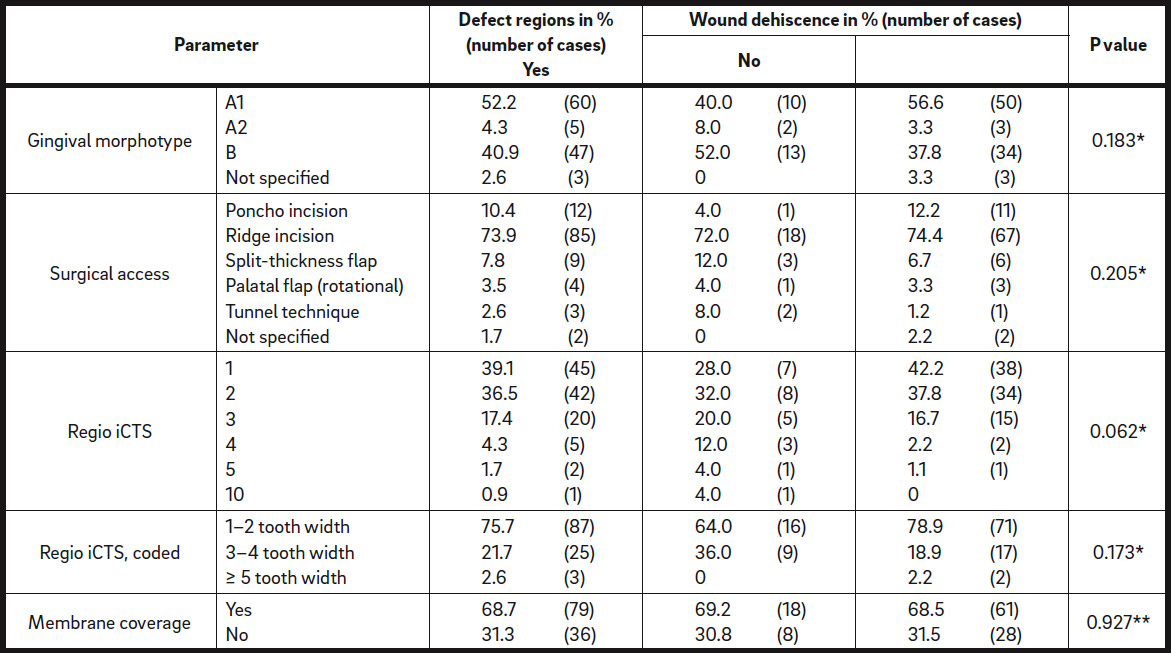
Of the 115 defect regions to be augmented, 72 were located in the maxilla (62.6%) and 43 in the mandible (37.4%; Table 2). Seventeen defect regions were horizontal (14.8%), 5 were vertical (4.3%) and the remaining 91 regions had a combined defect type (79.1%). For 2 cases, the defect type was not recorded. Sixty of the defect regions presented with thin gingival morphotype A1 (52.2%), 5 with thin gingival morphotype A2 (4.3%) and 47 with thick gingival morphotype B (40.9%; Table 2).
The following surgical approaches were used to assess the defect sites: In 85 cases, ridge incision (73.9%); in 12 cases, poncho incision (10.4%); in 9 cases, split-thickness flap (7.8%); in 4 cases, palatal flap (3.5%); and in 3 cases, tunnel technique (2.6%; Table 2). In 2 cases, the surgical approach was not documented. For ridge augmentation, a mixture of autogenous bone and DBBM was used in 106 defect regions (92.2%) and other materials in 8 defect regions (6.9%), and in 1 defect region (0.9%), the material used was not documented. The iCTS was covered with a native bilayer collagen membrane in 79 of the defect regions (68.7%) or left uncovered in 36 cases (31.3%; Table 2).
Distribution of defect regions in maxillae and mandibles. Total number of defect regions: n = 115.
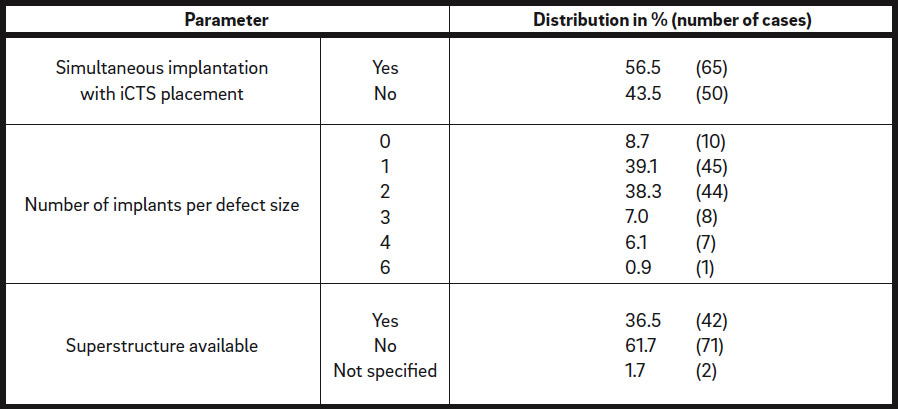
Depending on the initial situation and potential for primary implant stability, implants were inserted simultaneously with iCTS placement in 65 cases (56.5%) or 5–8 months after augmentation in 50 defect regions (43.5%; Table 3). A superstructure was available in 42 defect regions (36.5%). Mainly 1 or 2 implants were inserted in each defect region (39.1% and 38.3%; Table 3).
Wound healing and exposure of iCTS
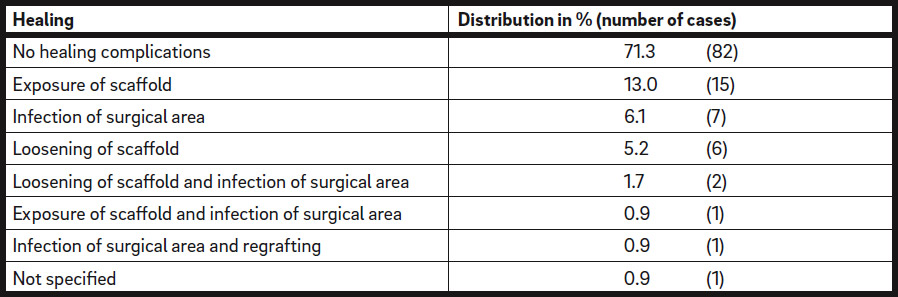
During the follow-up period of 6 months until re-entry, no healing complications were observed in 82 of the defect regions (71.3%; Table 4a). For the other 33 defects, the following healing complications were identified: 15 cases with iCTS exposure (13.0%), 7 cases with postoperative infection of the surgical area (6.1%) and 6 cases with loosening of the iCTS (5.2%). In 3 of the defect regions, exposure or loosening of the iCTS, together with postoperative infection, was documented (2.6%; Table 4a). Premature removal of exposed iCTSs was not necessary in any of the cases. In 1 defect region, healing complications were not specified (0.9%). One augmented site without any signs of dehiscence had to be regrafted owing to postoperative infection (0.9%; Table 4a).
Healing process. Total number of defect regions: n = 115.
Postoperative complications. Total number of defect regions: n = 115.
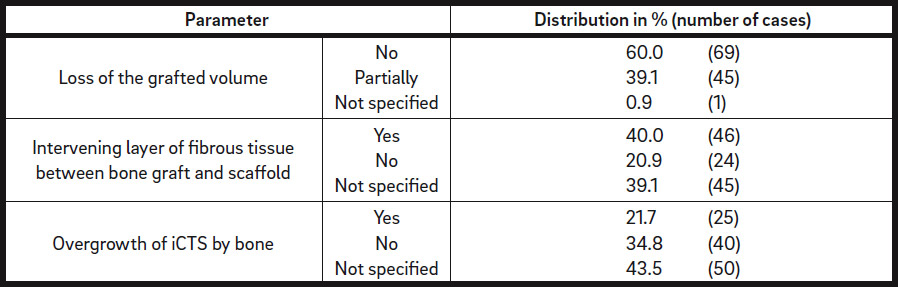
Six months postsurgery, all of the augmented regions showed sufficiently grafted volume. Staged implant placement was possible in all of the cases. The following postoperative complications were documented: Minor loss of the grafted volume in 45 cases (39.1%) was regrafted at re-entry when deemed necessary, an intervening layer of fibrous tissue between the bone graft and scaffold was observed in 46 defects (40.0%), and overgrowth of the scaffold by bone occurred in 25 regions (21.7%; Table 4b).
Wound dehiscence
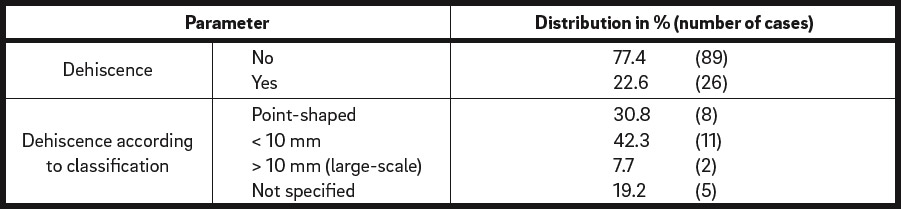
Overall, 26 of the 115 defect regions developed wound dehiscence (22.6%), while 89 did not (77.4%; Table 5a). According to wound dehiscence classification, wound dehiscence was point-shaped in 8 of the cases (30.8%), < 10 mm in 11 of the cases (42.3%) and > 10 mm in 2 of the cases (7.7%). In 5 of the cases with dehiscence, the classification of wound dehiscence was not specified (19.2%).
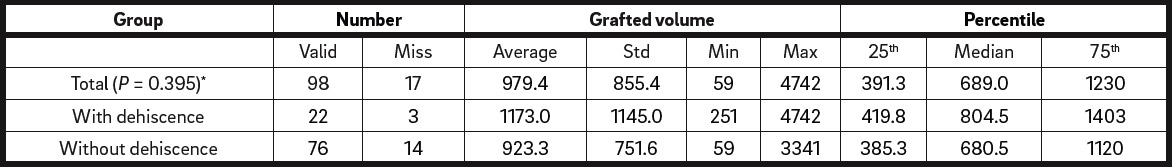
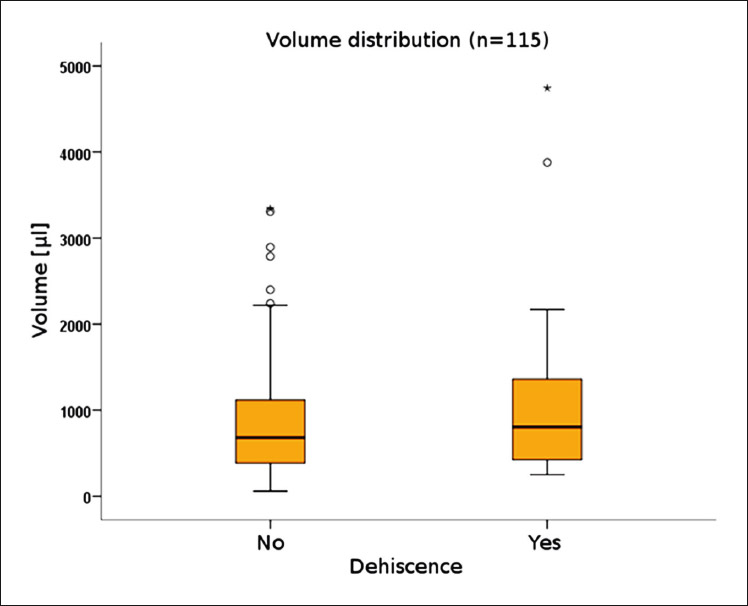
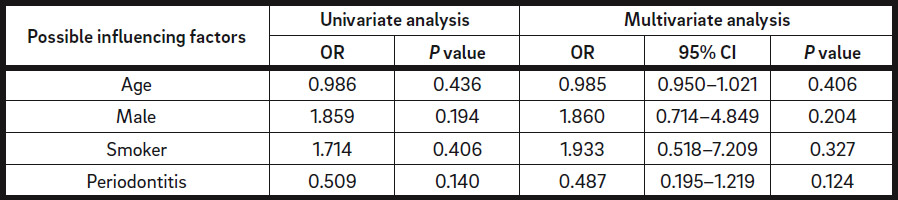
The grafted volume in the defect regions with dehiscence (1,173 ± 1,145 μL) was not statistically different from that in the regions without dehiscence (923.3 ± 751.6 μL; P = 0.395; Fig. 2 & Table 5b). Surgery-related parameters, including gingival morphotype (P = 0.183), surgical access (P = 0.205), membrane coverage (P = 0.927) and regio iCTS, coded (P = 0.173), did not show significant association with the prevalence of dehiscence (Table 5c). However, it should be noted, that a tendency to higher prevalence of dehiscence with mesiodistal width of the defect (regio iCTS: P = 0.062; Table 5c) was observed, but statistical significance was not achieved. No association of wound dehiscence with demographic (age, sex) or potential risk factors (smoking, periodontal disease) was found, as proved by different statistical approaches (Tables 6a & b).
Discussion
Prediction and improvement of factors influencing the healing process and treatment outcome are of great importance in implant dentistry.20Giglio J, Laskin D. Perioperative errors contributing to implant failure. → Oral Maxillofac Surg Clin North Am. 1998;2:197–202. In the present study, the application of an iCTS with DBBM and autogenous bone was found to result in sufficient grafted volumes and satisfactory clinical outcome. Prevalence of dehiscence was not affected by the demographic or surgical parameters analyzed. The presence of dehiscence did not influence the augmentation volume or implant insertion.
In the present study, an iCTS, together with a mixture of DBBM and autogenous bone, was mainly used (in 92.2% of defects) for complex alveolar bone augmentation. Typical complications, including infections and dehiscence, were easily treated. Dehiscence did not affect the final outcome, since augmented volumes were not affected and implant insertion was possible in all of the cases. These results are in line with previous studies showing similar effectiveness of iCTSs in the healing process21Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. that is comparable with that of custom-made titanium scaffolds.22Rasia-dal Polo M, Poli PP, Rancitelli D, Beretta M, Maiorana C. Alveolar ridge reconstruction with titanium meshes: a systematic review of the literature. → Med Oral Patol Oral Cir Bucal. 2014 Nov;19(6):e639–46. 23Sagheb K, Schiegnitz E, Moergel M, Walter C, Al-Nawas B, Wagner W. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. → Int J Implant Dent. 2017 Dec;3(1):36. doi: 10.1186/ s40729-017-0097-z. Application of an iCTS, together with a mixture of DBBM and autogenous bone, was previously shown to result in sufficient augmented volume and good clinical outcome.24Sagheb K, Schiegnitz E, Moergel M, Walter C, Al-Nawas B, Wagner W. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. → Int J Implant Dent. 2017 Dec;3(1):36. doi: 10.1186/ s40729-017-0097-z. Although 7 out of 21 cases showed exposure after 5–12 weeks, grafting was successful in all of the cases and implant survival was 100% after mean follow-up of 12 months.25Sagheb K, Schiegnitz E, Moergel M, Walter C, Al-Nawas B, Wagner W. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. → Int J Implant Dent. 2017 Dec;3(1):36. doi: 10.1186/ s40729-017-0097-z. No negative impact of dehiscence on the clinical outcome was found. Another study compared custom-made titanium devices with conventional titanium scaffolds for alveolar bone augmentation in 26 patients.26Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. In this study, mucosal rupture was observed less frequently with the use of custom-made titanium scaffolds (in 1 patient, 7.7%) in comparison with application of the conventional titanium devices (in 3 patients, 23.1%), but the difference was not statistically significant. However, the operation time was significantly shorter and the number of retaining screws used significantly fewer in the custom-made group than in the commercial titanium device group. Taken together, application of custom-made titanium scaffolds is associated with fewer complications and shorter operation time.
Different factors have been shown to have an impact on the success of dental procedures. To the best of our knowledge, the association of wound dehiscence with various demographic and surgical parameters was investigated for the first time in our retrospective analysis. Important influencing factors, including age, sex, smoking, periodontitis, gingival morphotype, surgical access, membrane coverage and regio iCTS, were taken into account. However, no statistically significant association between the prevalence of dehiscence with demographic or surgical parameters was found. A tendency to greater dehiscence with regio iCTS might point to increasing prevalence of dehiscence with the width of the defect. One possible reason could be the greater disturbance of blood supply in larger defects that impairs optimal wound closure. However, this hypothesis needs further clinical and experimental analysis. Application of native collagen membranes did not show increased prevalence of membrane exposure or wound dehiscence.27Becker J, Al-Nawas B, Klein MO, Schliephake H, Terheyden H, Schwarz F. Use of a new cross-linked collagen membrane for the treatment of dehiscence-type defects at titanium implants: a prospective, randomized-controlled double-blinded clinical multicenter study. → Clin Oral Implants Res. 2009 Jul;20(7):742–9. 28Tal H, Kozlovsky A, Artzi Z, Nemcovsky CE, Moses O. Cross-linked and non-cross-linked collagen barrier membranes disintegrate following surgical exposure to the oral environment: a histological study in the cat. → Clin Oral Implants Res. 2008 Aug;19(8):760–6.
Wound dehiscence. Total number of defect regions: n = 115.
Grafted volume is similar in defects with dehiscence to those without dehiscence. Total number of defect regions: n = 115.
Distribution of augmented volumes was not statistically different between defect sites with and without wound dehiscence, as calculated by the Mann–Whitney U test. Total number of defect sites: n = 115.
An important advantage of iCTSs is the easy handling and the perfect fitting of the scaffolds, resulting in fewer injuries and shorter operation time. In contrast, conventional titanium scaffolds need to be adapted during surgery, necessitating time-consuming cutting and bending of the scaffolds.29Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. 30Ciocca L, Ragazzini S, Fantini M, Corinaldesi G, Scotti R. Work flow for the prosthetic rehabilitation of atrophic patients with a minimal-intervention CAD/CAM approach. → J Prosthet Dent. 2015 Jul;114(1):22–6.
Effects of surgical parameters on wound dehiscence. Total number of defect regions: n = 115.
Distribution of possible influencing factors. Total number of defect regions: n = 115.
Logistic regression analysis on possible influencing factors. Total number of defect regions: n = 115.
An evidence-based review on clinical results in alveolar ridge augmentation showed that an average horizontal and vertical volume gain of 3.7 mm is possible using particulate guided bone regeneration techniques.31Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. These results can be significantly improved by inlay or onlay bone grafts using extraoral bone blocks or by distraction osteogenesis. However, these techniques seem to be accompanied by a higher complication rate, that is, infection and loss of block grafts subsequent to exposure.32Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. By using titanium mesh in combination with particulate grafts, the user is able to perform larger-sized bone grafting, and the technique appears to be much more forgiving of exposure. The results of our retrospective study are in strong accordance with these results.
Our study has some limitations. The data were analyzed retrospectively; thus, information regarding postoperative complications and some surgical parameters was missing in a few cases. The clinical outcome of iCTSs was not compared with that of conventional titanium scaffolds or in combination with other augmentation techniques. This would be of interest for future studies. Therefore, further prospective long-term and randomized controlled clinical trials in larger patient cohorts are of interest to provide more evidence for improved clinical outcomes using the iCTS in comparison with other techniques.
Conclusion
The results of this study suggest that application of an iCTS with an equal mixture of autogenous bone and DBBM offers a reliable grafting technique with low sensitivity to wound dehiscence. Prevalence of dehiscence was not influenced by the demographic or surgical parameters analyzed.
Competing interests
Data analysis by an independent statistician was supported by a grant from Geistlich Pharma AG, Switzerland. The authors declare that they have no other competing interests.
Ethical approval and informed consent
This retrospective study was performed without any further consequences for the patient. According to this and the hospital laws of the individual states (Krankenhauslandesgesetz von RLP), no approval by the local ethics committee is necessary. Furthermore, for this type of study, formal consent is not required.
References
| 1. | ↑ | Barker D. Implant assessment. → Dent Update. 2012 Mar;39(2):128–32, 134. |
| 2. | ↑ | Moraschini V, Poubel LA, Ferreira VF, Barboza Edos S. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: a systematic review. → Int J Oral Maxillofac Surg. 2015 Mar;44(3):377–88. |
| 3. | ↑ | Buser D, Martin W, Belser UC. Optimizing esthetics for implant restorations in the anterior maxilla: anatomic and surgical considerations. → Int J Oral Maxillofac Implants. 2004;19 Suppl:43–61. |
| 4, 7, 22. | ↑ | Rasia-dal Polo M, Poli PP, Rancitelli D, Beretta M, Maiorana C. Alveolar ridge reconstruction with titanium meshes: a systematic review of the literature. → Med Oral Patol Oral Cir Bucal. 2014 Nov;19(6):e639–46. |
| 5, 31, 32. | ↑ | Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. |
| 6, 15. | ↑ | Hämmerle CH, Araújo MG, Simion M; Osteology Consensus Group 2011. Evidence-based knowledge on the biology and treatment of extraction sockets. → Clin Oral Implants Res. 2012 Feb;23 Suppl 5:80–2. |
| 8. | ↑ | Maiorana C, Santoro F, Rabagliati M, Salina S. Evaluation of the use of iliac cancellous bone and anorganic bovine bone in the reconstruction of the atrophic maxilla with titanium mesh: a clinical and histologic investigation. → Int J Oral Maxillofac Implants. 2001 May–Jun;16(3):427–32. |
| 9. | ↑ | Artzi Z, Dayan D, Alpern Y, Nemcovsky CE. Vertical ridge augmentation using xenogenic material supported by a configured titanium mesh: clinicohistopathologic and histochemical study. → Int J Oral Maxillofac Implants. 2003 May–Jun;18(3):440–6. |
| 10. | ↑ | Troeltzsch M, Troeltzsch M, Kauffmann P, Gruber R, Brockmeyer P, Moser N, Rau A, Schliephake H. Clinical efficacy of grafting materials in alveolar ridge augmentation: a systematic review. → J Craniomaxillofac Surg. 2016 Oct;44(10):1618–29. |
| 11, 21. | ↑ | Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. |
| 12, 14, 30. | ↑ | Ciocca L, Ragazzini S, Fantini M, Corinaldesi G, Scotti R. Work flow for the prosthetic rehabilitation of atrophic patients with a minimal-intervention CAD/CAM approach. → J Prosthet Dent. 2015 Jul;114(1):22–6. |
| 13, 26, 29. | ↑ | Sumida T, Otawa N, Kamata YU, Kamakura S, Matsushita T, Kitagaki H, Mori S, Sasaki K, Fujibayashi S, Takemoto M, Yamaguchi A, Sohmura T, Nakamura T, Mori Y. Custom-made titanium devices as membranes for bone augmentation in implant treatment: clinical application and the comparison with conventional titanium mesh. → J Craniomaxillofac Surg. 2015 Dec;43(10):2183–8. |
| 16. | ↑ | Maiorana C, Santoro F, Rabagliati M, Salina S. Evaluation of the use of iliac cancellous bone and anorganic bovine bone in the reconstruction of the atrophic maxilla with titanium mesh: a clinical and histologic investigation. → Int J Oral Maxillofac Implants. 2001 May–Jun;16(3):427–32. |
| 17, 23, 24, 25. | ↑ | Sagheb K, Schiegnitz E, Moergel M, Walter C, Al-Nawas B, Wagner W. Clinical outcome of alveolar ridge augmentation with individualized CAD-CAM-produced titanium mesh. → Int J Implant Dent. 2017 Dec;3(1):36. doi: 10.1186/ s40729-017-0097-z. |
| 18. | ↑ | Müller HP, Eger T. Gingival phenotypes in young male adults. → J Clin Periodontol. 1997 Jan;24(1):65–71. |
| 19. | ↑ | Hoexter DL, Epstein SB. The Poncho Flap (repositioned perforated attached gingival flap). → Oral Implantol. 1975 Spring;5(4):547–52. |
| 20. | ↑ | Giglio J, Laskin D. Perioperative errors contributing to implant failure. → Oral Maxillofac Surg Clin North Am. 1998;2:197–202. |
| 27. | ↑ | Becker J, Al-Nawas B, Klein MO, Schliephake H, Terheyden H, Schwarz F. Use of a new cross-linked collagen membrane for the treatment of dehiscence-type defects at titanium implants: a prospective, randomized-controlled double-blinded clinical multicenter study. → Clin Oral Implants Res. 2009 Jul;20(7):742–9. |
| 28. | ↑ | Tal H, Kozlovsky A, Artzi Z, Nemcovsky CE, Moses O. Cross-linked and non-cross-linked collagen barrier membranes disintegrate following surgical exposure to the oral environment: a histological study in the cat. → Clin Oral Implants Res. 2008 Aug;19(8):760–6. |






Leave a Reply
Be the First to Comment!